Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
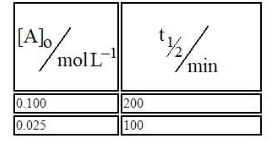
For a given reaction $$R\rightarrow P,t_{1/2}$$ is related to $$[A]_{\circ}$$ as given in table. Given: $$\log 2=0.30$$ Which of the following is true? A. The order of the reaction is 1/2.B.If $$[A]_{\circ}$$ is 1 M, then $$t_{1/2}$$ is $$200\sqrt{10}min$$ C.The order of the reaction changes to 1 if the concentration of reactant changes from 0.100 M to 0.500 M . D. $$t_{1/2}$$ is 800 min for $$[A]_{\circ}=1.6M$$ Choose the correct answer from the options given below: Options
Create a FREE account and get:
Educational materials for JEE preparation
