Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following molecules/species:
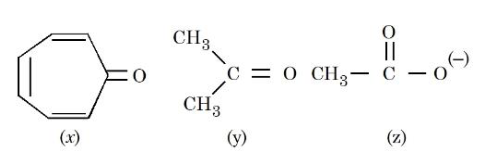
The correct order of carbon-oxygen double bond length is:
The length of a C=O bond depends mainly on its bond order. A higher bond order means stronger overlap, larger electron density between the nuclei and hence a shorter bond. Conversely, a lower bond order leads to a longer bond.
Bond order is reduced whenever the π-electrons of the C=O group are delocalised through resonance or conjugation.
Case (y): acetone
Acetone contains an isolated carbonyl group with no π-conjugation. There is essentially one dominant structure
$$CH_3-CO-CH_3$$
so the C=O bond order is almost exactly 2. Therefore its C=O bond is the shortest of the three molecules considered.
Case (z): acetate ion, $$CH_3COO^-$$
For the carboxylate ion two equivalent resonance forms exist:
$$CH_3-C(=O)O^- \;\;\;\; \leftrightarrow \;\;\;\; CH_3-C(O^-) = O$$
Because both structures contribute equally, each C-O bond has a bond order of $$\frac{1+2}{2}=1.5$$. A bond order of 1.5 is much lower than 2, so the C-O bonds in acetate are considerably longer than the C=O bond in acetone.
Case (x): tropone-like compound
Tropone is $$\text{cyclohepta-2,4,6-trien-1-one}$$. The carbonyl double bond is conjugated with three C=C bonds in the 7-membered ring, producing resonance structures such as
$$\text{O}^--C^+$$ delocalised over the ring.
This delocalisation removes part of the double-bond character from the C=O bond, lowering its bond order to a value between 1.5 (acetate) and 2 (acetone). Consequently its C=O bond length lies between those of acetate and acetone.
Putting the three results together:
• acetate ion (z): bond order 1.5 → longest C-O bond
• tropone-like compound (x): bond order <2 but >1.5 → intermediate length
• acetone (y): bond order ≈2 → shortest C=O bond
Therefore the correct order is
$$\text{z} \; \gt \; \text{x} \; \gt \; \text{y}$$
Option C which is: z > x > y
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
