Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following reactions. From these reactions which reaction will give carboxylic acid as a major product ?
(A) $$R-C \equiv N \xrightarrow[\text{mild condition}]{(i) H^+/H_2O}$$
(B) $$R-MgX \xrightarrow[(ii) H_3O^+]{(i) CO_2}$$
(C) $$R-C \equiv N \xrightarrow[(ii) H_3O^+]{(i) SnCl_2/HCl}$$
(D) $$R-CH_2-OH \xrightarrow{PCC}$$
(E)
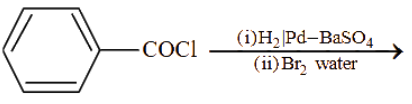
Choose the correct answer from the options given below :
Create a FREE account and get:
Educational materials for JEE preparation
