Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the decomposition of azomethane $$CH_3N_2CH_3(g) \rightarrow CH_3CH_3(g) + N_2$$, a first order reaction, the variation in partial pressure with time at $$600 \text{ K}$$ is given as shown.
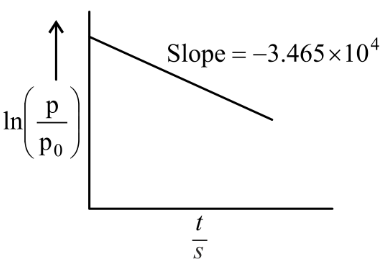
The half life of the reaction is ______ $$\times 10^{-5}$$ s.
Correct Answer: 2
To find the half-life of the first-order decomposition of azomethane, we start by determining the rate constant (k) from the provided graph. For a first-order reaction, the integrated rate equation relating partial pressure to time is ln(p/p₀) = -kt.
When you plot ln(p/p₀) on the y-axis against time (t) on the x-axis, the relationship mirrors the equation of a straight line (y = mx) where the slope (m) is equal to -k.
The given graph indicates that the slope is -3.465 × 10⁴. By setting this equal to -k, we determine that the rate constant is k = 3.465 × 10⁴ s⁻¹.
Next, we apply the standard half-life formula for a first-order reaction, which is t₁/₂ = 0.693 / k.
Substituting our calculated value for k into the formula gives: t₁/₂ = 0.693 / (3.465 × 10⁴)
Dividing 0.693 by 3.465 gives 0.2. Therefore, the equation simplifies to: t₁/₂ = 0.2 × 10⁻⁴ s
To match the format requested in the question, we rewrite this in standard scientific notation: t₁/₂ = 2 × 10⁻⁵ s
The right answer is 2.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
