Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A mixture containing the following four compounds is extracted with 1 M HCl. The compound that goes to aqueous layer is:
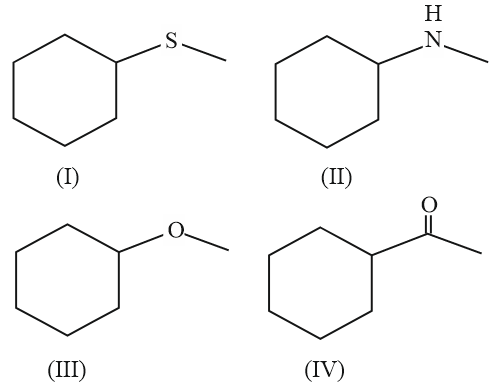
Extraction with 1 M HCl separates compounds based on their basicity. Basic compounds react with HCl to form water-soluble ammonium salts, which partition into the aqueous layer.
Among the given compounds, only compound (II) contains a basic amine group. It reacts with HCl to form the corresponding ammonium salt:
$$R_2NH\ +\ HCL\ \longrightarrow\ R_2N^+Cl^-$$
This salt is soluble in water and therefore transfers to the aqueous layer.
The other compounds (thioether, ether, and ketone) are non-basic and remain in the organic layer.
Create a FREE account and get:
Educational materials for JEE preparation
