Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
$$2NO(g) + Cl_2(g) \rightleftharpoons 2NOCl(s)$$
This reaction was studied at -10°C and the following data was obtained:
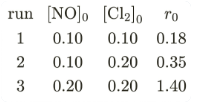
$$[NO]_{\circ}$$ and $$[Cl_{2}]_{\circ}$$ are the initial concentrations and $$r_{\circ}$$ is the initial reaction rate. The overall order of the reaction is ________. (Round off to the Nearest Integer).
Correct Answer: 3
The rate law has the form $$r = k[\text{NO}]^a[\text{Cl}_2]^b$$. We determine the orders $$a$$ and $$b$$ from the experimental data.
Comparing Run 1 and Run 2, where $$[\text{NO}]$$ is held constant at 0.10 and $$[\text{Cl}_2]$$ doubles from 0.10 to 0.20: $$\frac{r_2}{r_1} = \frac{0.35}{0.18} \approx 1.944 \approx 2^b$$. Since $$2^1 = 2 \approx 1.944$$, the order with respect to $$\text{Cl}_2$$ is $$b \approx 1$$.
Comparing Run 2 and Run 3, where $$[\text{Cl}_2]$$ is held constant at 0.20 and $$[\text{NO}]$$ doubles from 0.10 to 0.20: $$\frac{r_3}{r_2} = \frac{1.40}{0.35} = 4.0 = 2^a$$. Since $$2^2 = 4$$, the order with respect to $$\text{NO}$$ is $$a = 2$$.
The overall order of the reaction is $$a + b = 2 + 1 = 3$$.
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.