Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
For the reaction $$2Fe^{3+}(aq) + 2I^-(aq) \to 2Fe^{2+}(aq) + I_2(s)$$, the magnitude of the standard molar free energy change, $$\Delta_r G_m^\circ$$ = -________ kJ (Round off to the Nearest Integer).
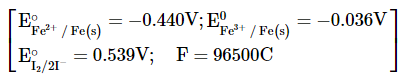
Correct Answer: 45
The overall reaction is
$$2Fe^{3+}(aq) + 2I^-(aq) \rightarrow 2Fe^{2+}(aq) + I_2(s)$$
Standard molar free-energy change is obtained from
$$\Delta_r G_m^\circ = -nF E_{\text{cell}}^\circ$$
Hence we need
1. $$E^\circ_{\text{cell}}$$ for the redox pair $$Fe^{3+}/Fe^{2+}$$ vs $$I_2/ I^-$$.
2. The electron count $$n$$ in the balanced ionic equation.
Case 1: Determining $$E^\circ_{Fe^{3+}/Fe^{2+}}$$
The data supplied are reduction potentials vs $$Fe(s)$$:
$$Fe^{3+} + 3e^- \rightarrow Fe(s) \;\;\;\; E_1^\circ = -0.036\; \text{V}$$
$$Fe^{2+} + 2e^- \rightarrow Fe(s) \;\;\;\; E_2^\circ = -0.440\; \text{V}$$
First convert each potential to its standard free energy:
$$\Delta G_1^\circ = -3F E_1^\circ = -3F(-0.036) = +0.108F$$
$$\Delta G_2^\circ = -2F E_2^\circ = -2F(-0.440) = +0.880F$$
Reverse the second half-reaction so that $$Fe(s)$$ cancels:
$$Fe(s) \rightarrow Fe^{2+} + 2e^- \;\;\;\; \Delta G^\circ = -\Delta G_2^\circ = -0.880F$$
Add this to the first reaction:
$$Fe^{3+} + 3e^- +\,Fe(s) \rightarrow Fe(s) + Fe^{2+} + 2e^-$$
which simplifies to
$$Fe^{3+} + e^- \rightarrow Fe^{2+}$$
The free-energy change for this step is
$$\Delta G_3^\circ = \Delta G_1^\circ - \Delta G_2^\circ = (0.108F) - (0.880F) = -0.772F$$
Therefore
$$E^\circ_{Fe^{3+}/Fe^{2+}} = -\dfrac{\Delta G_3^\circ}{F} = 0.772\; \text{V}$$
Case 2: Cell potential for the required reaction
Half-reactions in the direction of the overall process:
Cathode (reduction): $$Fe^{3+} + e^- \rightarrow Fe^{2+}\;\; E_c^\circ = 0.772\; \text{V}$$
Anode (oxidation): $$2I^- \rightarrow I_2 + 2e^-$$
(The given reduction potential is $$I_2 + 2e^- \rightarrow 2I^- \;\; E^\circ = 0.539\; \text{V}$$, so for oxidation we take $$-0.539\; \text{V}$$.)
Hence
$$E_{\text{cell}}^\circ = E_c^\circ - E_a^\circ = 0.772\; \text{V} - 0.539\; \text{V} = 0.233\; \text{V}$$
Case 3: Free-energy change
The balanced ionic equation transfers $$n = 2$$ electrons (compare either half-reaction). Using $$F = 96500\; \text{C mol}^{-1}$$:
$$\Delta_r G_m^\circ = -nF E_{\text{cell}}^\circ$$
$$\Delta_r G_m^\circ = -2 \times 96500 \times 0.233$$
$$\Delta_r G_m^\circ = -4.497 \times 10^4\; \text{J mol}^{-1}$$
Convert to kilojoules:
$$\Delta_r G_m^\circ \approx -44.97\; \text{kJ mol}^{-1}$$
Rounded to the nearest integer, the magnitude is $$\boxed{45\ \text{kJ}}$$.
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.