Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements : Statement I: The rate law for the reaction $$A + B \rightarrow C$$ is rate $$(r) = k[A]^2[B]$$. When the concentration of both A and B is doubled, the reaction rate is increased "$$x$$" times. Statement II :
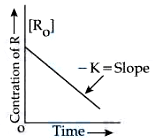
The figure is showing "the variation in concentration against time plot" for a "$$y$$" order reaction. The Value of $$x + y$$ is ______
Correct Answer: 8
If concentration of A and B are doubled then x= (2^2)[A]*(2)[B] = 8[A][B], x=8
Since the curve between concentration and time is linear it is a zero order reaction. Hence, y =0
x+y = 8
Create a FREE account and get:
Educational materials for JEE preparation
