Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
How many compounds among the following compounds show inductive, mesomeric as well as hyperconjugation effects?
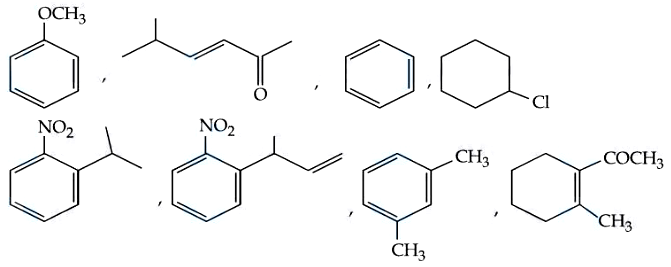
Correct Answer: 4
Create a FREE account and get:
Educational materials for JEE preparation
