Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements :
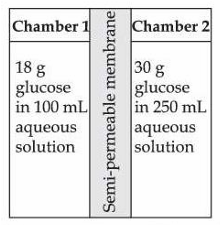
Statement I : H$$_2$$O molecules move from the chamber 1 to chamber 2.
Statement II : The osmotic pressure of a solution prepared by dissolving 50 mg of potassium sulphate (molar mass = 174 g/mol) in 2 L of water (at 27 °C) is 0.0107 bar. (Given: R = 0.083 dm$$^3$$ bar K$$^{-1}$$ mol$$^{-1}$$ and assume complete dissociation of electrolyte)
In the light of the above statements, choose the correct answer from the options given below :
Create a FREE account and get:
Educational materials for JEE preparation
