Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
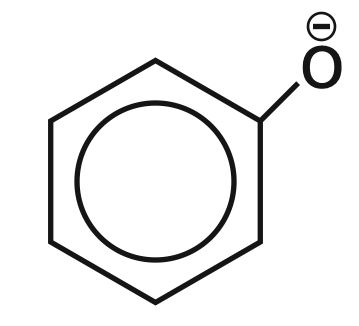
Which one of the following substituents at para-position is most effective in stabilizing the phenoxide ion?
$$-CH_3$$ (Option A): This is an electron-donating group through the $$+I$$ (inductive) effect and hyperconjugation. It destabilizes the phenoxide ion.
$$-OCH_3$$ (Option B): It can donate into the ring via the $$+M$$ (mesomeric) effect. This makes it a strong electron-donating group that destabilizes the ion.
$$-COCH_3$$ (Option C): This acetyl group is a strong electron-withdrawing group. It exerts both a $$-I$$ effect and a powerful $$-M$$ (resonance withdrawing) effect.
Because it is at the para-position, the negative charge from the phenoxide oxygen can be delocalized through the ring directly onto the oxygen of the carbonyl group. This is the most effective form of stabilization.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.