Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The increasing order of the reactivity of the following halides for the S$$_{N}$$1 reaction is
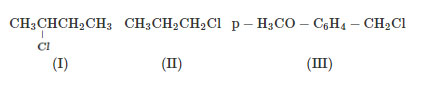
In an $$S_N1$$ reaction, reactivity depends on the stability of the carbocation formed: $$3^\circ > 2^\circ > 1^\circ$$
(II) is a $$1^\circ$$ alkyl halide, forming an unstable primary carbocation → least reactive.
(I) is a $$2^\circ$$ alkyl halide, forming a more stable secondary carbocation.
(III) is a benzylic halide. The carbocation formed is highly stabilized by resonance and the $$+M$$ effect of the $$-OCH_3$$ group.
Therefore, the increasing order of $$S_N1$$ reactivity is: $$(II)<(I)<(III)$$
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.