Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
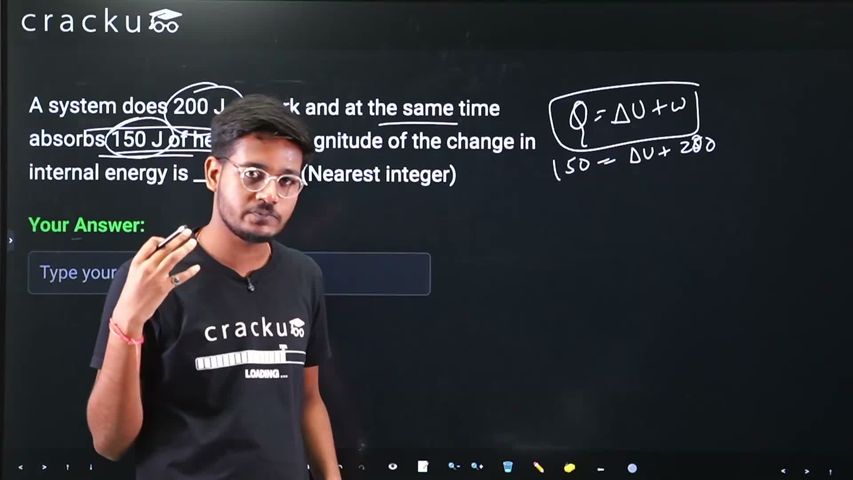
A system does 200 J of work and at the same time absorbs 150 J of heat. The magnitude of the change in internal energy is _________J. (Nearest integer)
Correct Answer: 50
We recall the First Law of Thermodynamics, which is stated as $$\Delta U = Q - W,$$ where $$\Delta U$$ is the change in internal energy of the system, $$Q$$ is the heat absorbed by the system from the surroundings, and $$W$$ is the work done by the system on the surroundings.
According to the question, the system absorbs heat, so the heat term is positive. Numerically we have
$$Q = +150\ \text{J}.$$
The system also does work on the surroundings, therefore the work term counted in the equation is also positive. The data give
$$W = +200\ \text{J}.$$
Now we substitute these two values into the First Law expression:
$$\Delta U = Q - W = (+150\ \text{J}) - (+200\ \text{J}).$$
Carrying out the subtraction step by step, we first write the numerical difference,
$$\Delta U = 150\ \text{J} - 200\ \text{J}.$$
Performing the arithmetic,
$$\Delta U = -50\ \text{J}.$$
The question asks for the magnitude of the change in internal energy, that is, the absolute value:
$$\left|\Delta U\right| = \left|-50\ \text{J}\right| = 50\ \text{J}.$$
So, the answer is $$50\ \text{J}.$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
