Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
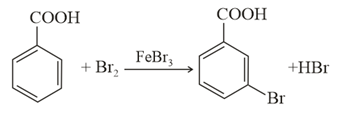
Consider the above reaction where 6.1 g of benzoic acid is used to get 7.8 g of m-bromo benzoic acid. The percentage yield of the product is ___.
(Round off to the Nearest integer)
[Given : Atomic masses : C = 12.0 u, H : 1.0 u, O : 16.0 u, Br = 80.0 u]
Correct Answer: 78
To determine the percentage yield, first calculate the molar masses of the reactant and product.
For benzoic acid:
$$\mathrm{C_7H_6O_2}$$
$$\mathrm{Molar\ Mass = (7\times12) + (6\times1) + (2\times16)}$$
$$\mathrm{= 84 + 6 + 32 = 122\ g\ mol^{-1}}$$
For m-bromobenzoic acid:
$$\mathrm{C_7H_5BrO_2}$$
$$\mathrm{Molar\ Mass = (7\times12) + (5\times1) + 80 + (2\times16)}$$
$$\mathrm{= 84 + 5 + 80 + 32 = 201\ g\ mol^{-1}}$$
The balanced reaction shows a $$\mathrm{1:1}$$ molar ratio between benzoic acid and m-bromobenzoic acid.
Moles of benzoic acid used:
$$\mathrm{Moles = \frac{6.1}{122}}$$
$$\mathrm{= 0.05\ mol}$$
Therefore,
$$\mathrm{0.05\ mol\ Benzoic\ Acid \rightarrow 0.05\ mol\ Product}$$
Theoretical yield of product:
$$\mathrm{Mass = Moles \times Molar\ Mass}$$
$$\mathrm{= 0.05 \times 201}$$
$$\mathrm{= 10.05\ g}$$
Actual yield obtained:
$$\mathrm{7.8\ g}$$
Percentage yield:
$$\mathrm{\%\ Yield = \frac{Actual\ Yield}{Theoretical\ Yield}\times100}$$
$$\mathrm{= \frac{7.8}{10.05}\times100}$$
$$\mathrm{= 77.61\%}$$
Rounding to the nearest integer:
$$\boxed{\mathrm{78}}$$
Create a FREE account and get:
Educational materials for JEE preparation
