Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements :
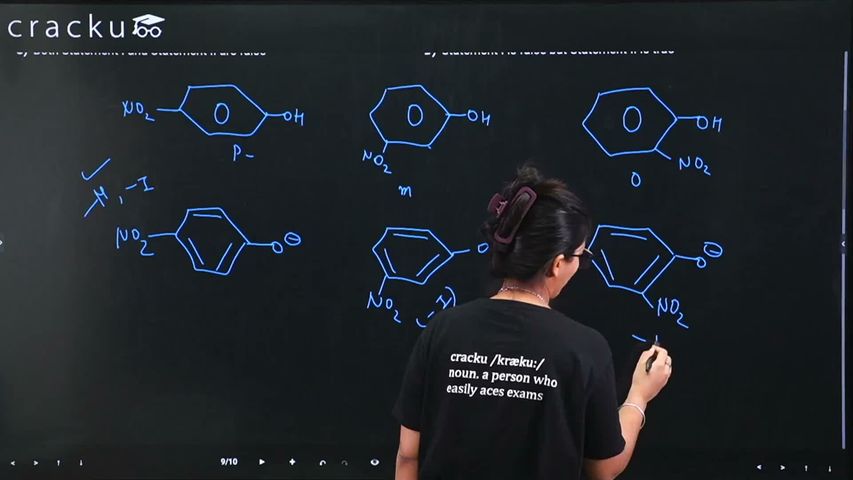
Statement (I) : p-nitrophenol is more acidic than m-nitrophenol and o-nitrophenol.
Statement (II) : Ethanol will give immediate turbidity with Lucas reagent.
In the light of the above statements, choose the correct answer from the options given below :
Statement I: p-Nitrophenol is more acidic than m-nitrophenol and o-nitrophenol.
The $$-NO_2$$ group is electron-withdrawing. At the para position, it stabilizes the phenoxide ion through resonance (direct conjugation), making p-nitrophenol the most acidic among the three isomers. This is true.
(Note: While o-nitrophenol has intramolecular hydrogen bonding which reduces its effective acidity in solution, p-nitrophenol has the strongest -R effect stabilization.)
Statement II: Ethanol will give immediate turbidity with Lucas reagent.
Lucas reagent (conc. HCl + ZnCl$$_2$$) is used to distinguish between primary, secondary, and tertiary alcohols. Primary alcohols like ethanol do not give immediate turbidity - they show no reaction at room temperature. Only tertiary alcohols give immediate turbidity. This is false.
Statement I is true but Statement II is false. The answer corresponds to Option (1).
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
