Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match List - I with List - II:
List - I (Chemical Reaction) List - II (Reagent used)
a. CH$$_3$$COOCH$$_2$$CH$$_3$$ $$\rightarrow$$ CH$$_3$$CH$$_2$$OH i. CH$$_3$$MgBr/H$$_3$$O$$^+$$ (one equivalent)
b. CH$$_3$$COOCH$$_3$$ $$\rightarrow$$ CH$$_3$$CHO ii. H$$_2$$SO$$_4$$/H$$_2$$O
c. CH$$_3$$C$$\equiv$$N $$\rightarrow$$ CH$$_3$$CHO iii. DIBAL-H/H$$_2$$O
d. iv. SnCl$$_2$$, HCl/H$$_2$$O
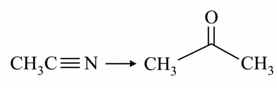
Choose the most appropriate match:
We look at transformation (a). The starting compound is $$\mathrm{CH_3COOCH_2CH_3}$$, an ester known as ethyl acetate. The desired product is $$\mathrm{CH_3CH_2OH}$$, ethanol. We recall the general hydrolysis reaction of an ester in acidic medium, which is stated as
$$$\mathrm{RCOOR' \;+\; H_2O \;\xrightarrow[\;]{H^+}\; RCOOH \;+\; R'OH}$$$
Here $$\mathrm{R = CH_3}$$ and $$\mathrm{R' = CH_2CH_3}$$, so hydrolysis will furnish acetic acid and ethanol. The reagent that supplies both water and acid is the mixture $$\mathrm{H_2SO_4/H_2O}$$, listed as (ii). Hence, for part (a) we must choose (ii).
Now we examine transformation (b). The substrate is $$\mathrm{CH_3COOCH_3}$$ (methyl acetate) and the target is $$\mathrm{CH_3CHO}$$ (acetaldehyde). Converting an ester directly to an aldehyde requires partial reduction that stops at the aldehyde stage. The reagent famous for this selective one-step reduction is DIBAL-H followed by aqueous work-up. The overall reaction may be represented as
$$$\mathrm{RCOOR' \;\xrightarrow[\;]{\text{DIBAL-H,}\; -78^{\circ}\text{C}} \; RCHO \;\xrightarrow{\;H_2O\;} RCHO}$$$
DIBAL-H/H$$_2$$O is option (iii), so (b) corresponds to (iii).
Next, consider transformation (c): $$\mathrm{CH_3C\equiv N \;\rightarrow\; CH_3CHO}$$. Converting a nitrile to an aldehyde is achieved by the Stephen reduction. The textbook Stephen reduction uses stannous chloride in the presence of hydrochloric acid, followed by hydrolysis:
$$$\mathrm{RCN \;+\; 2\,SnCl_2 \;+\; 2\,HCl \;\xrightarrow[\;]{\;} RCH=NH\cdot HCl \;\xrightarrow{\;H_2O\;} RCHO}$$$
This reagent set is given as (iv) $$\mathrm{SnCl_2,\,HCl/H_2O}$$. Therefore, (c) matches (iv).
Finally, we look at transformation (d): $$\mathrm{CH_3C\equiv N \;\rightarrow\; CH_3COCH_3}$$, i.e. conversion of acetonitrile to acetone. A single equivalent of a Grignard reagent adds an alkyl group to the electrophilic carbon of the nitrile, giving an imine magnesium salt; hydrolysis of the imine then produces a ketone. The general formula is
$$$\mathrm{RCN \;+\; R''MgBr \;\xrightarrow[\;]{\;} RC(R'')=NMgBr \;\xrightarrow{\;H_3O^+\;} RC(O)R''}$$$
With $$\mathrm{R = CH_3}$$ and $$\mathrm{R'' = CH_3}$$, we indeed obtain $$\mathrm{CH_3COCH_3}$$. The required reagent is $$\mathrm{CH_3MgBr/H_3O^+}$$, listed as (i). Thus, (d) corresponds to (i).
Collecting all the matches we have:
$$$\text{(a)} \rightarrow (ii), \qquad \text{(b)} \rightarrow (iii), \qquad \text{(c)} \rightarrow (iv), \qquad \text{(d)} \rightarrow (i)$$$
This sequence appears exactly in Option A.
Hence, the correct answer is Option A.
Create a FREE account and get:
Educational materials for JEE preparation
