Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Considering the above reaction, X and Y respectively are: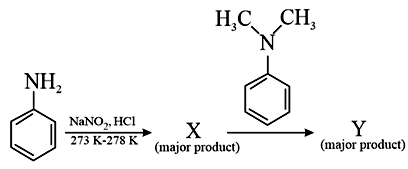
An aromatic primary amine first reacts with $$NaNO_2/HCl$$ at $$0{-}5^{\circ}{\rm C}$$ to give a diazonium salt. This transformation is called diazotisation.
General equation of diazotisation:
$$Ar{-}NH_2 + NaNO_2 + 2\,HCl \; (0{-}5^{\circ}{\rm C}) \;\longrightarrow\; Ar{-}N_2^{+}Cl^{-} + NaCl + 2\,H_2O$$
Thus, for aniline $$\big( C_6H_5NH_2 \big)$$,
$$C_6H_5NH_2 \xrightarrow{NaNO_2/HCl,\;0{-}5^{\circ}{\rm C}} C_6H_5N_2^{+}Cl^{-}$$
so compound $$X$$ is benzene diazonium chloride.
The diazonium group can be replaced by $$CN$$ using the Sandmeyer reaction with $$CuCN$$.
Sandmeyer reaction (for $$CN$$ replacement):
$$Ar{-}N_2^{+}Cl^{-} + CuCN \;\longrightarrow\; Ar{-}CN + N_2 \uparrow + CuCl$$
Applying it to $$X$$:
$$C_6H_5N_2^{+}Cl^{-} \xrightarrow{CuCN} C_6H_5CN + N_2 \uparrow + CuCl$$
Hence compound $$Y$$ is benzonitrile (cyanobenzene).
Therefore, $$X = C_6H_5N_2^{+}Cl^{-}\;(\text{benzene diazonium chloride})$$ and $$Y = C_6H_5CN\;(\text{benzonitrile}).$$
These correspond to Option B (given as answer number 2). Hence the correct choice is Option B.
Educational materials for JEE preparation
