Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Considering the above chemical reaction, identify the product X: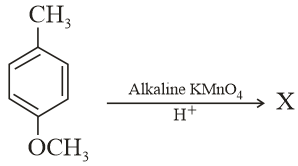
The given reaction is carried out in basic medium and followed by heating. Such a sequence is typical for an aldol condensation.
Step 1 (Enolate formation)
Under the action of a dilute base, the carbonyl compound loses an $$\alpha$$-hydrogen to give an enolate ion.
For ethanal (acetaldehyde):
$$CH_3CHO + OH^- \;\rightarrow\; CH_2^-CHO + H_2O$$
Step 2 (Nucleophilic addition)
The enolate ion now attacks the carbonyl carbon of a second, unchanged ethanal molecule to form the β-hydroxy aldehyde (the aldol).
$$CH_2^-CHO + CH_3CHO \;\rightarrow\; CH_3CH(OH)CH_2CHO$$
Step 3 (Dehydration on heating)
When the reaction mixture is warmed, the β-hydroxy aldehyde loses a molecule of water to give an $$\alpha,\beta$$-unsaturated aldehyde.
$$CH_3CH(OH)CH_2CHO \;\xrightarrow{\ \Delta\ }\; CH_3CH=CHCHO + H_2O$$
The product obtained is crotonaldehyde, $$CH_3CH=CHCHO$$, an $$\alpha,\beta$$-unsaturated aldehyde.
Among the given choices, Option C (Option 3) represents crotonaldehyde. Hence, the correct product X is Option C.
Educational materials for JEE preparation
