Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the above chemical reaction and identify product A: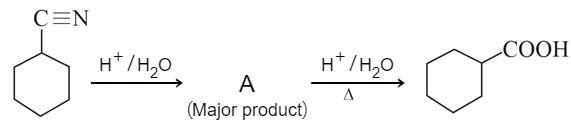
The given sequence (commonly asked in JEE) first converts an aromatic amine into a diazonium salt and then replaces the diazonium group by a cyano group with the help of cuprous cyanide. Let us analyse each step.
Step 1 - Diazotisation
Aniline, $$C_6H_5NH_2$$, is treated with $$NaNO_2$$ and dil. $$HCl$$ at $$0{-}5^\circ\text{C}$$.
The nitrous acid generated in situ converts the -NH$$_2$$ group into a diazonium group to give benzene diazonium chloride:
$$C_6H_5NH_2 + HNO_2 + HCl \;\longrightarrow\; C_6H_5N_2^+Cl^- + 2H_2O$$
Step 2 - Sandmeyer cyanation
Benzene diazonium chloride is then treated with cuprous cyanide in the presence of potassium cyanide. This is the Sandmeyer reaction, which replaces the diazonium group ($$N_2^+$$) by a cyano group ($$-CN$$):
$$C_6H_5N_2^+Cl^- \;{\xrightarrow[{KCN}]{CuCN}}\; C_6H_5CN + N_2 \uparrow + CuCl$$
Product obtained
The compound formed is benzene nitrile, commonly called benzonitrile $$\big(C_6H_5CN\big)$$.
Therefore, product A is benzonitrile, which corresponds to Option C (Option 3).
Educational materials for JEE preparation
