Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following complex ions $$P = [FeF_6]^{3-}$$, $$Q = [V(H_2O)_6]^{2+}$$, $$R = [Fe(H_2O)_6]^{2+}$$. The correct order of the complex ions, according to their spin only magnetic moment values (in B.M.) is :
First recall the spin-only magnetic moment formula for any transition-metal complex:
$$\mu_{\text{spin}} = \sqrt{n(n+2)}\; \text{B.M.}$$
where $$n$$ is the number of unpaired $$d$$-electrons.
We therefore need, for each complex ion,
(i) the oxidation state and hence the $$d$$-electron count of the metal,
(ii) whether the complex is high-spin or low-spin (decided by ligand field strength).
Case 1 : $$P = [\,FeF_6\,]^{3-}$$
Oxidation state of Fe: $$x + 6(-1) = -3 \;\Rightarrow\; x = +3$$ so $$Fe^{3+}$$.
Electronic configuration of $$Fe^{3+}$$: $$[Ar]\;3d^5$$ (five $$d$$-electrons).
Fluoride ($$F^-$$) is a weak-field ligand, so the octahedral complex is high-spin. Hence no pairing occurs and all five $$d$$-electrons remain unpaired.
Therefore $$n_P = 5$$ and $$\mu_P = \sqrt{5(5+2)} = \sqrt{35} \approx 5.92\;\text{B.M.}$$
Case 2 : $$Q = [\,V(H_2O)_6\,]^{2+}$$
Oxidation state of V: $$x + 6(0) = +2 \;\Rightarrow\; x = +2$$ so $$V^{2+}$$.
Electronic configuration of $$V^{2+}$$: $$[Ar]\;3d^3$$ (three $$d$$-electrons).
Water ($$H_2O$$) is also a weak-field ligand, giving a high-spin octahedral complex. All three electrons stay unpaired.
Thus $$n_Q = 3$$ and $$\mu_Q = \sqrt{3(3+2)} = \sqrt{15} \approx 3.87\;\text{B.M.}$$
Case 3 : $$R = [\,Fe(H_2O)_6\,]^{2+}$$
Oxidation state of Fe: $$x + 6(0) = +2 \;\Rightarrow\; x = +2$$ so $$Fe^{2+}$$.
Electronic configuration of $$Fe^{2+}$$: $$[Ar]\;3d^6$$ (six $$d$$-electrons).
Again, $$H_2O$$ is a weak-field ligand ⇒ high-spin. The high-spin octahedral $$d^6$$ configuration ($$t_{2g}^4e_g^2$$) contains four unpaired electrons.
Hence $$n_R = 4$$ and $$\mu_R = \sqrt{4(4+2)} = \sqrt{24} \approx 4.90\;\text{B.M.}$$
Collecting the magnetic moments:
$$\mu_Q \approx 3.87\; \text{B.M.} \lt \mu_R \approx 4.90\; \text{B.M.} \lt \mu_P \approx 5.92\; \text{B.M.}$$
Therefore the required ascending order is
$$Q \lt R \lt P$$
Which corresponds to Option C.
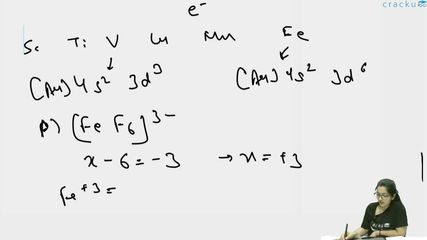
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
