Tollens' reagent (ammoniacal silver nitrate, $[Ag(NH_3)_2]^+$) is a mild oxidizing agent prepared in a basic solution.
- Positive Test (Forms Silver Mirror): It successfully oxidizes aldehydes into carboxylic acid salts, reducing the silver ions into solid metallic silver (the "mirror"). It also reacts with hemiacetals (because they open up to form aldehydes in solution) and $\alpha$-hydroxy ketones.
- Negative Test (No Silver Mirror): It is not strong enough to oxidize ketones, standard alcohols, or acetals/ketals.
Because Tollens' reagent is basic, any compound capable of tautomerizing (shifting between a keto and enol form) will rapidly do so in the solution. We must evaluate the most stable form of each molecule.
Analysis of the Compounds
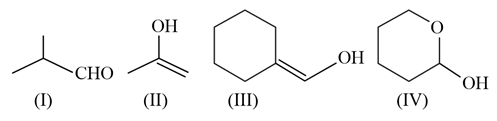
- Compound (I): Isobutyraldehyde
- Structure: This molecule clearly contains a free aldehyde group ($-CHO$).
- Reaction: Since it is a standard aliphatic aldehyde, it will readily be oxidized.
- Result: Forms a silver mirror.
- Compound (II): Prop-1-en-2-ol
- Structure: This is an enol (an alkene and an alcohol on the same carbon).
- Reaction: Enols are generally unstable compared to their carbonyl counterparts. In the basic medium of Tollens' reagent, this molecule will rapidly undergo tautomerization to its more stable keto form, which is acetone ($CH_3-CO-CH_3$). Acetone is a ketone. Since ketones cannot be oxidized by Tollens' reagent, no reaction occurs.
- Result: Does NOT form a silver mirror.
- Compound (III): (Cyclohexylidene)methanol
- Structure: This is also an enol (the double bond is right next to the $-OH$ group).
- Reaction: Just like Compound II, this will tautomerize in the basic Tollens' solution. However, when the double bond shifts into the ring and the proton moves to the carbon, it forms cyclohexanecarbaldehyde (a cyclohexane ring attached to a $-CHO$ group). Because its keto form is an aldehyde, it will successfully react with the reagent.
- Result: Forms a silver mirror.
- Compound (IV): 2-Hydroxytetrahydropyran
- Structure: This is a cyclic hemiacetal (a carbon bonded to both an $-OH$ group and an $-OR$ ether linkage within a ring).
- Reaction: In an aqueous solution, cyclic hemiacetals exist in a dynamic equilibrium with their open-chain forms. When this ring opens, it forms 5-hydroxypentanal, revealing a free aldehyde group at the end of the chain. This free aldehyde will then react with the Tollens' reagent.
- Result: Forms a silver mirror.
Conclusion
Because it tautomerizes into a ketone rather than an aldehyde, Compound (II) is the only molecule in this lineup that will not give a positive Tollens' test.