Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given
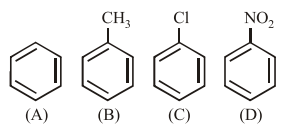
In the above compounds correct order of reactivity in electrophilic substitution reactions will be:
Electrophilic aromatic substitution reactivity depends on electron density, where electron-donating groups (EDGs) activate the ring and electron-withdrawing groups (EWGs) deactivate it.
$$-CH_3$$ (B) activates via hyperconjugation, while $$-Cl$$ (C) and $$-NO_2$$ (D) deactivate the ring through $$-I$$ and $$-M$$ effects respectively.
Consequently, toluene (B) is the most reactive followed by benzene (A), with nitrobenzene (D) being the least reactive due to strong deactivation.
The correct order of reactivity is B > A > C > D
Create a FREE account and get:
Educational materials for JEE preparation
