Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
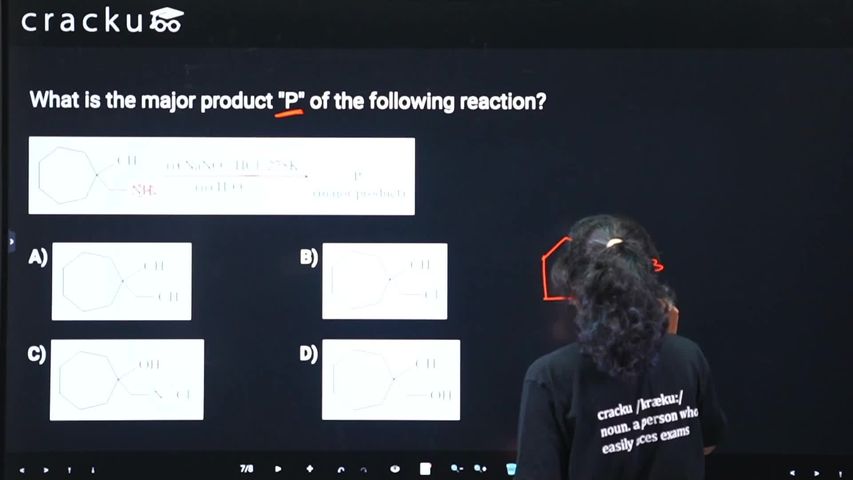
In the general aliphatic reaction, when a primary aliphatic amine ($$R-NH_2$$) is treated with nitrous acid ($$HNO_2$$)—which is generated in situ by mixing sodium nitrite ($$NaNO_2$$) and hydrochloric acid ($$HCl$$)—it rapidly undergoes diazotization to form an aliphatic diazonium salt intermediate ($$[R-N_2^+Cl^-]$$). Unlike aromatic diazonium salts, these aliphatic diazonium salts are highly unstable even at very low temperatures. Nitrogen gas is an excellent leaving group and immediately departs, allowing the resulting carbocation to be instantly attacked by water ($$H_2O$$) from the aqueous solvent. This transformation is represented by the general equation:
$$R-NH_2 \xrightarrow{NaNO_2 + HCl} [R-N_2^+Cl^-] \xrightarrow{H_2O} R-OH + N_2 \uparrow + HCl$$
Consequently, as shown in the final step of the reaction sequence, the major compound formed is an aliphatic alcohol ($$R-OH$$), accompanied by the brisk effervescence of nitrogen gas ($$N_2$$). Therefore, this corresponds to Option D.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
