Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the given plot of enthalpy of the following reaction between A and B.
A + B $$\rightarrow$$ C + D. Identify the incorrect statement.
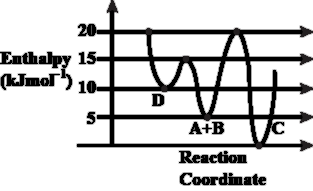
First let us recall the definition of activation enthalpy. For a given elementary step, the activation enthalpy is the difference in enthalpy between the reactants and the top of the energy barrier (the transition state). Symbolically we write
$$E_a = H_{\text{TS}} - H_{\text{reactants}}$$
where $$H_{\text{TS}}$$ is the enthalpy of the transition state and $$H_{\text{reactants}}$$ is the enthalpy of the reacting species at that point in the mechanism.
According to the diagram supplied in the question (enthalpy on the vertical axis and reaction progress on the horizontal axis) there are two possible pathways that start from the common reactant ensemble $$A+B$$ and end in the two alternative products $$C$$ and $$D$$. The salient numerical features of the plot can be stated in words:
• Product $$C$$ lies lower on the enthalpy scale than product $$D$$. Hence we have
$$H_C < H_D$$
so that the formation of $$C$$ is thermodynamically more favourable.
• The peak (transition state) on the pathway leading to $$D$$ is lower than the peak on the pathway leading to $$C$$. Writing the two activation enthalpies as $$E_{a,D}$$ and $$E_{a,C}$$, the diagram clearly shows
$$E_{a,D} < E_{a,C}$$
In fact, the numerical vertical difference between the two peaks is labelled as $$5\ \text{kJ mol}^{-1}$$, but the higher peak belongs to the route that forms $$C$$, not the one that forms $$D$$. Quantitatively:
$$E_{a,C}=E_{a,D}+5\ \text{kJ mol}^{-1}$$
Combining the last two statements gives us the strict inequality
$$E_{a,C} > E_{a,D}$$
Now we analyse each option with these relations in mind.
Option A. “Formation of A and B from C has highest enthalpy of activation.” To go backward from $$C$$ to $$A+B$$, the system must climb from the low level $$H_C$$ up to the higher transition state situated on the $$A+B \to C$$ curve. Since $$H_C$$ is the lowest point on the entire diagram, this backward climb is indeed the largest uphill journey shown. Hence Option A is a correct statement.
Option B. “D is kinetically stable product.” Kinetic stability (greater rate of formation) is governed by the magnitude of $$E_a$$; the smaller the activation enthalpy, the faster the product forms. Because $$E_{a,D} < E_{a,C}$$, product $$D$$ forms faster and is therefore the kinetically favoured product. Option B is therefore correct.
Option C. “C is the thermodynamically stable product.” Thermodynamic stability depends on the final enthalpy level. Since $$H_C < H_D$$, product $$C$$ is indeed more stable in a thermodynamic sense. So Option C is correct.
Option D. “Activation enthalpy to form C is 5 kJ mol$$^{-1}$$ less than that to form D.” We have already established the quantitative relation $$E_{a,C}=E_{a,D}+5\ \text{kJ mol}^{-1}$$, which is algebraically equivalent to
$$E_{a,C}-E_{a,D}=+5\ \text{kJ mol}^{-1}$$
or, rearranged,
$$E_{a,C} > E_{a,D}$$
This means the activation enthalpy for $$C$$ is larger, not smaller, by exactly $$5\ \text{kJ mol}^{-1}$$. Therefore the wording in Option D directly contradicts the numerical data of the diagram, making Option D an incorrect statement.
Since the problem asks us to identify the incorrect statement, we must select Option D.
Hence, the correct answer is Option 4.
Create a FREE account and get:
Educational materials for JEE preparation
