Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Consider the following volume-temperature (V-T) diagram for the expansion of 5 moles of an ideal monoatomic gas.
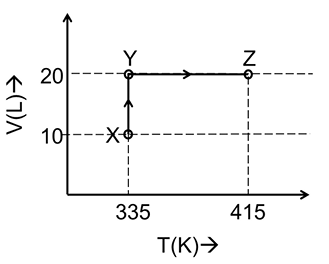
Considering only P-V work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $$X \rightarrow Y \rightarrow Z$$ is ______.
[Use the given data: Molar heat capacity of the gas for the given temperature range, $$C_{V,m} = 12$$ J $$K^{-1}$$ $$mol^{-1}$$ and gas constant, $$R = 8.3$$ J $$K^{-1}$$ $$mol^{-1}$$]
Correct Answer: 8120
For an ideal gas the molar enthalpy depends only on its temperature: $$H = C_{p,m}\,T$$. Hence the enthalpy change for any multi-step process depends only on the temperatures of the initial and final states, not on the path followed.
The V-T diagram supplied in the question gives the coordinates of the three states (all volumes are in litres, temperatures in kelvin):
• State $$X : (V = 2\,\text{L},\;T = 400\,\text{K})$$
• State $$Y : (V = 4\,\text{L},\;T = 400\,\text{K})$$ (isothermal expansion $$X \rightarrow Y$$)
• State $$Z : (V = 4\,\text{L},\;T = 480\,\text{K})$$ (isochoric heating $$Y \rightarrow Z$$)
Thus the overall temperature change for the complete transformation $$X \rightarrow Y \rightarrow Z$$ is
$$\Delta T \;=\; T_Z \;-\; T_X \;=\; 480\ \text{K} \;-\; 400\ \text{K} \;=\; 80\ \text{K}$$
The given data are
• number of moles, $$n = 5$$
• molar heat capacity at constant volume, $$C_{V,m} = 12\,\text{J K}^{-1}\text{ mol}^{-1}$$
• gas constant, $$R = 8.3\,\text{J K}^{-1}\text{ mol}^{-1}$$
For an ideal gas, $$C_{p,m} = C_{V,m} + R$$, therefore
$$C_{p,m} = 12 + 8.3 = 20.3\;\text{J K}^{-1}\text{ mol}^{-1}$$
The total enthalpy change is then
$$\Delta H \;=\; n\,C_{p,m}\,\Delta T$$ $$\Delta H \;=\; 5 \times 20.3 \times 80$$ $$\Delta H \;=\; 101.5 \times 80$$ $$\Delta H \;=\; 8120\ \text{J}$$
Hence, the total change in enthalpy for the given transformation is 8120 J.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation
