Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The option(s) with correct sequence of reagents for the conversion of P to Q is(are)
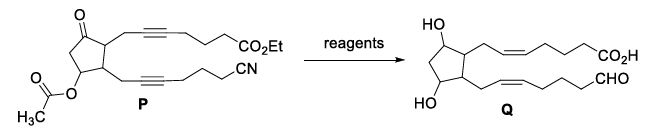
Three different reducible groups are present in $$P$$: an internal alkyne, a nitro group and a carbonyl group. They have to be converted successively to a cis-alkene, an amine and an alcohol in the final product $$Q$$. For each transformation we choose the most chemoselective reagent and decide the safest order so that the later steps do not undo or over-reduce what has already been achieved.
Step 1 - Reduction of the C≡C bond only
Lindlar’s catalyst with $$H_2$$ stops at the cis-alkene stage and does not disturb either nitro or carbonyl groups.
Hence the first reagent must be “Lindlar’s catalyst, $$H_2$$”.
Step 2 - Reduction of the −NO2 group
The most selective reagent that converts $$-NO_2$$ to $$-NH_2$$ in the presence of a carbonyl is $$SnCl_2/HCl$$.
Using it before reducing the carbonyl is preferable because the strongly acidic medium would protonate or even dehydrate an alcohol if it were already present.
Therefore the second reagent should be “$$SnCl_2/HCl$$”.
Step 3 - Reduction of the C=O group
After the nitro group has become an amine, an electrically neutral hydride reagent that works in neutral or mildly basic medium is required for the carbonyl.
$$NaBH_4$$ cleanly converts aldehydes/ketones to alcohols and leaves alkenes and amines untouched.
Thus the third reagent is “$$NaBH_4$$”.
Step 4 - Acidic work-up
The hydride reduction is performed in methanol or ethanol; an aqueous acid work-up ($$H_3O^+$$) is necessary to protonate the alkoxide, giving the free alcohol and completing the synthesis.
Consequently the fourth reagent is “$$H_3O^+$$”.
Why the other sequences fail
• If $$NaBH_4$$ comes before $$SnCl_2/HCl$$ (Options C and D), the alcohol formed in step 1 would face strongly acidic conditions in step 2 and might dehydrate or undergo side reactions.
• Placing $$H_3O^+$$ immediately after the partial hydrogenation (Option B) serves no purpose and the subsequent acidic reduction with $$SnCl_2/HCl$$ would again endanger any alcohol already present.
• Any sequence that applies $$SnCl_2/HCl$$ or $$NaBH_4$$ before Lindlar’s hydrogenation risks over-reduction of the alkyne (because later catalytic hydrogenation over Pd/C would reduce the newly formed alkene all the way to an alkane).
Thus the only logically consistent sequence is
i) Lindlar's catalyst, $$H_2$$ → ii) $$SnCl_2/HCl$$ → iii) $$NaBH_4$$ → iv) $$H_3O^+$$
Option A matches this order.
Final Answer: Option A which is: i) Lindlar's catalyst, $$H_2$$; ii) $$SnCl_2$$/HCl; iii) $$NaBH_4$$; iv) $$H_3O^+$$.
Create a FREE account and get:
Educational materials for JEE preparation
