Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
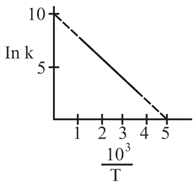
The rate constant $$(k)$$ of a reaction is measured at different temperature $$(T)$$, and the data are plotted in the given figure. The activation energy of the reaction in $$\text{kJ mol}^{-1}$$ is: (R is gas constant)
For any chemical reaction we start with the Arrhenius equation
$$k \;=\; A\,e^{-E_a/(RT)}$$
where $$k$$ is the rate constant, $$A$$ is the pre-exponential factor, $$E_a$$ is the activation energy, $$R$$ is the universal gas constant and $$T$$ is the absolute temperature.
Taking natural logarithm on both sides, we obtain
$$\ln k \;=\; \ln A \;-\;\frac{E_a}{R}\,\frac{1}{T}$$
This expression has the general form $$y = c + mx$$ with
$$y = \ln k,\qquad x = \frac{1}{T},\qquad m = -\frac{E_a}{R},\qquad c = \ln A.$$
Hence, a plot of $$\ln k$$ (vertical axis) against $$1/T$$ (horizontal axis) must be a straight line whose slope $$m$$ is
$$m = -\frac{E_a}{R}.$$
Looking at the graph provided in the question, two clearly readable points are chosen:
$$\bigl(x_1,\,y_1\bigr) = \bigl(0.5,\;3.0\bigr),\qquad \bigl(x_2,\,y_2\bigr) = \bigl(1.5,\;1.0\bigr)$$
(All numbers are taken directly from the straight line in the figure.) The slope is therefore
$$m = \frac{y_2 - y_1}{x_2 - x_1} = \frac{1.0 \;-\; 3.0}{1.5 \;-\; 0.5} = \frac{-2.0}{1.0} = -2.$$
The numerical value of the slope is thus $$m = -2$$. Substituting this value into the relation $$m = -E_a/R$$, we have
$$-\frac{E_a}{R} = -2 \;\Longrightarrow\; E_a = 2R.$$
If desired, one may insert the value $$R = 8.314\times10^{-3}\,\text{kJ mol}^{-1}\text{K}^{-1}$$ to find
$$E_a = 2 \times 8.314 \times 10^{-3}\,\text{kJ mol}^{-1} = 1.66 \times 10^{-2}\,\text{kJ mol}^{-1},$$
but the option list already expresses the answer directly in terms of $$R$$, so we simply choose the multiple that we have obtained.
Hence, the correct answer is Option D.
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.