Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The correct stability order of the following resonance structures of $$CH_3 - CH = CH - CHO$$ i
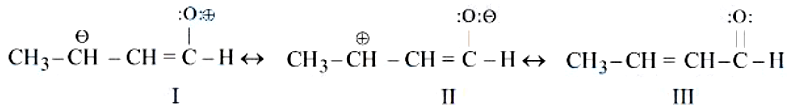
According to rules of making resonance structures, the most stable resonance structure is one which will have no charge, next would be the structure in which electronegative atom gets a negative charge i.e. Oxygen must have negative charge. The least stable resonating structure would be the structure that has a positive charge on oxygen opposite to its tendency. Hence, III>II>I
Create a FREE account and get:
Educational materials for JEE preparation
