Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the List-I with List-II.
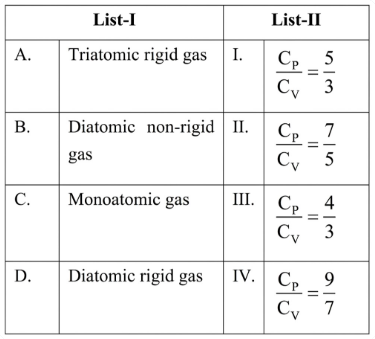
Choose the correct answer from the options given below :
For an ideal gas molecule the ratio of specific heats is given by the kinetic-theory formula
$$\gamma = \frac{C_p}{C_v} = 1 + \frac{2}{f}$$
where $$f$$ is the total number of degrees of freedom of one molecule.
Case A : Triatomic rigid gas
A rigid triatomic molecule that is non-linear (e.g. $$H_2O$$) has
• $$3$$ translational degrees + $$3$$ rotational degrees $$\Rightarrow f = 6$$.
Therefore $$\gamma = 1 + \frac{2}{6} = 1 + \frac13 = \frac43$$.
So A matches List-II item III.
Case B : Diatomic non-rigid gas
A non-rigid diatomic molecule possesses vibration along the bond in addition to translation and rotation.
• Translational : $$3$$, Rotational : $$2$$, Vibrational : $$2$$ (one kinetic + one potential)
Hence $$f = 3 + 2 + 2 = 7$$.
Thus $$\gamma = 1 + \frac{2}{7} = \frac{9}{7}$$.
So B matches List-II item IV.
Case C : Monoatomic gas
A monoatomic molecule can only translate.
• $$3$$ translational degrees, no rotation or vibration $$\Rightarrow f = 3$$.
Hence $$\gamma = 1 + \frac{2}{3} = \frac{5}{3}$$.
So C matches List-II item I.
Case D : Diatomic rigid gas
A rigid diatomic molecule has no vibrational contribution at ordinary temperatures.
• Translational : $$3$$, Rotational : $$2$$ $$\Rightarrow f = 5$$.
Therefore $$\gamma = 1 + \frac{2}{5} = \frac{7}{5}$$.
So D matches List-II item II.
Collecting the correspondences:
A - III, B - IV, C - I, D - II
This is exactly Option A.
Final answer: Option A.
Educational materials for JEE preparation
