Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Match the LIST-I with LIST-II.
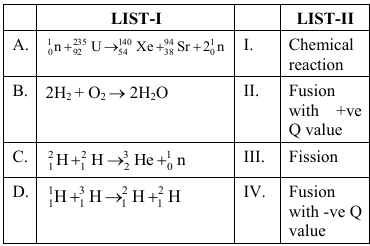
Choose the correct answer from the options given below :
First recognise the four classes given in LIST-II.
I Chemical reaction - involves only electrons, no change in nucleus, energy of the order of $$\text{kJ mol}^{-1}$$.
II Fusion with $$+$$ve Q value - two light nuclei combine, total mass decreases, energy is released ($$Q \gt 0$$).
III Fission - a heavy nucleus splits into two medium-mass nuclei and a few neutrons, energy is released ($$Q \gt 0$$).
IV Fusion with $$-$$ve Q value - two nuclei combine but the product mass is larger, energy is absorbed ($$Q \lt 0$$).
Case A: $$^{1}_{0}n + {}^{235}_{92}U \rightarrow {}^{140}_{54}Xe + {}^{94}_{38}Sr + 2\,^{1}_{0}n$$
A single heavy nucleus $$^{235}U$$ breaks into two medium nuclei plus neutrons. According to the definition, this is nuclear fission. Hence A → III.
Case B: $$2H_2 + O_2 \rightarrow 2H_2O$$
Only the electronic configuration of atoms changes; nuclei remain unchanged. This is an ordinary chemical reaction. Hence B → I.
Case C: $$^{2}_{1}H + {}^{2}_{1}H \rightarrow {}^{3}_{2}He + {}^{1}_{0}n$$
Two deuterons fuse to form a helium-3 nucleus and a neutron.
Mass of reactants: $$M_r = 2 \times 2.014102\,u = 4.028204\,u$$.
Mass of products: $$M_p = 3.016029\,u + 1.008665\,u = 4.024694\,u$$.
$$\Delta M = M_r - M_p = 0.003510\,u \gt 0$$, so $$Q = \Delta M c^2 \approx 3.27\ \text{MeV} \gt 0$$.
Energy is released; it is a fusion reaction with positive Q value. Hence C → II.
Case D: $$^{1}_{1}H + {}^{3}_{1}H \rightarrow {}^{2}_{1}H + {}^{2}_{1}H$$
Mass of reactants: $$1.007825\,u + 3.016049\,u = 4.023874\,u$$.
Mass of products: $$2 \times 2.014102\,u = 4.028204\,u$$.
$$\Delta M = M_r - M_p = -0.004330\,u \lt 0$$, so $$Q = \Delta M c^2 \approx -4.03\ \text{MeV} \lt 0$$.
Energy is absorbed; it is a fusion reaction with negative Q value. Hence D → IV.
Thus the correct matching is
A - III, B - I, C - II, D - IV.
The option representing this pattern is Option B.
Final Answer: Option B
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.