Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: [6] Annulene, [8] Annulene and cis-[10] Annulene are respectively aromatic, not-aromatic and aromatic.
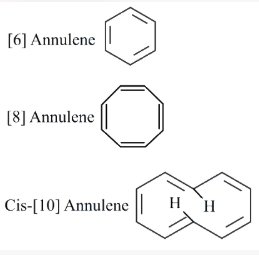
Reason R: Planarity is one of the requirements of aromatic systems.
In the light of the above statements, choose the most appropriate answer from the options given below.
We need to evaluate the Assertion and Reason about annulenes and aromaticity.
Assertion A: [6] Annulene, [8] Annulene and cis-[10] Annulene are respectively aromatic, not-aromatic and aromatic.
Recall Huckel's Rule: A compound is aromatic if it is cyclic, planar, fully conjugated, and has $$(4n + 2)$$ pi electrons.
Analysis of each annulene:
[6] Annulene (Benzene): Has 6 pi electrons. Using $$(4n + 2)$$ with $$n = 1$$: $$4(1) + 2 = 6$$. It is cyclic, planar, and fully conjugated. So it is aromatic. This part is correct.
[8] Annulene (Cyclooctatetraene): Has 8 pi electrons. $$8 = 4n$$ with $$n = 2$$, so it has $$4n$$ electrons (anti-aromatic if planar). However, it adopts a tub-shaped (non-planar) geometry to avoid anti-aromaticity. So it is non-aromatic. This part is correct.
cis-[10] Annulene (all-cis cyclodecapentaene): Has 10 pi electrons. $$4n + 2 = 10$$ with $$n = 2$$. While it satisfies Huckel's rule electronically, the all-cis configuration of [10] annulene is not planar due to angle strain and steric interactions between interior hydrogen atoms. Since it is not planar, it is not aromatic.
Therefore, Assertion A is incorrect because cis-[10] annulene is not aromatic (it fails the planarity requirement).
Reason R: "Planarity is one of the requirements of aromatic systems."
This is correct. Planarity is indeed a necessary condition for aromaticity to allow continuous overlap of p-orbitals.
Conclusion: A is not correct but R is correct.
The correct answer is Option D.
Create a FREE account and get:
Educational materials for JEE preparation
