Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
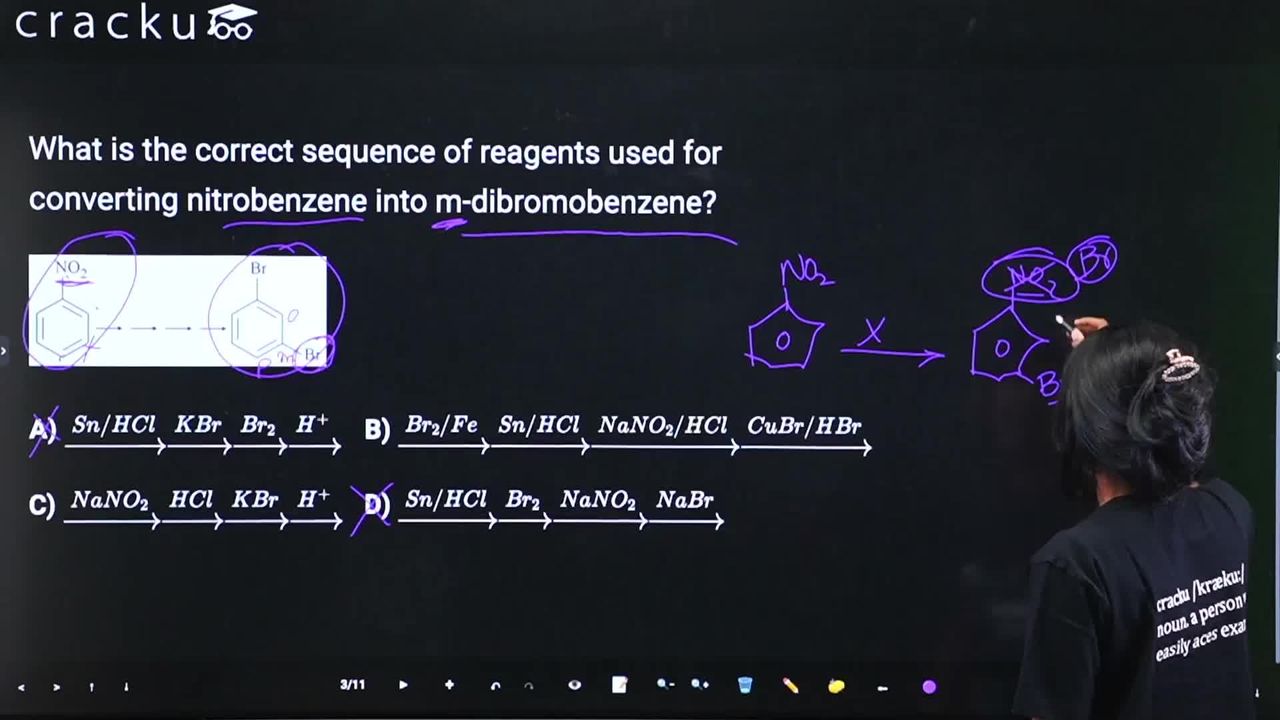
What is the correct sequence of reagents used for converting nitrobenzene into m-dibromobenzene?
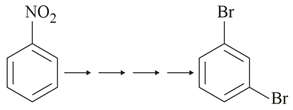
Using $Br_2$ and an iron catalyst ($Fe$ or $FeBr_3$), a bromine atom is added to the ring. Because the $-NO_2$ group is a meta-director, the bromine attaches at the meta position, securing the required 1,3 relationship.
$$C_6H_5NO_2 \xrightarrow{Br_2/Fe} m\text{-}Br-C_6H_4-NO_2$$
Tin ($Sn$) and hydrochloric acid ($HCl$) act as a reducing agent to convert the nitro group ($-NO_2$) into a primary amine group ($-NH_2$), forming $m$-bromoaniline.
$$m\text{-}Br-C_6H_4-NO_2 \xrightarrow{Sn/HCl} m\text{-}Br-C_6H_4-NH_2$$
Reacting the primary aromatic amine with cold sodium nitrite and hydrochloric acid ($NaNO_2/HCl$) converts the amine into a highly reactive diazonium salt.
$$m\text{-}Br-C_6H_4-NH_2 \xrightarrow{NaNO_2/HCl} m\text{-}Br-C_6H_4-N_2^+Cl^-$$
Finally, treating the diazonium salt with cuprous bromide and hydrobromic acid ($CuBr/HBr$) displaces the diazonium group (releasing nitrogen gas) and replaces it with a second bromine atom, yielding the final product.
$$m\text{-}Br-C_6H_4-N_2^+Cl^- \xrightarrow{CuBr/HBr} m\text{-dibromobenzene}$$
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.