Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
We need to identify the products formed when $$KMnO_4$$ is heated at 513 K (along with $$O_2$$).
Write the thermal decomposition reaction.
When potassium permanganate is heated, it undergoes the following decomposition:
$$2KMnO_4 \xrightarrow{\Delta} K_2MnO_4 + MnO_2 + O_2$$
Verify the balancing.
Left side: K: 2, Mn: 2, O: 8
Right side: K: 2, Mn: 1+1=2, O: 4+2+2=8. Balanced.
Understand the chemistry.
In $$KMnO_4$$, manganese is in the +7 oxidation state. During thermal decomposition:
- One Mn goes from +7 to +6 in $$K_2MnO_4$$ (potassium manganate) - this is a reduction
- Another Mn goes from +7 to +4 in $$MnO_2$$ (manganese dioxide) - this is also a reduction
- Oxygen goes from -2 to 0 in $$O_2$$ - this is an oxidation
This is a disproportionation-type reaction where the oxygen is oxidised while manganese is reduced to two different lower oxidation states.
The products (along with $$O_2$$) are $$K_2MnO_4$$ and $$MnO_2$$.
The correct answer is Option (4): $$K_2MnO_4$$ and $$MnO_2$$.
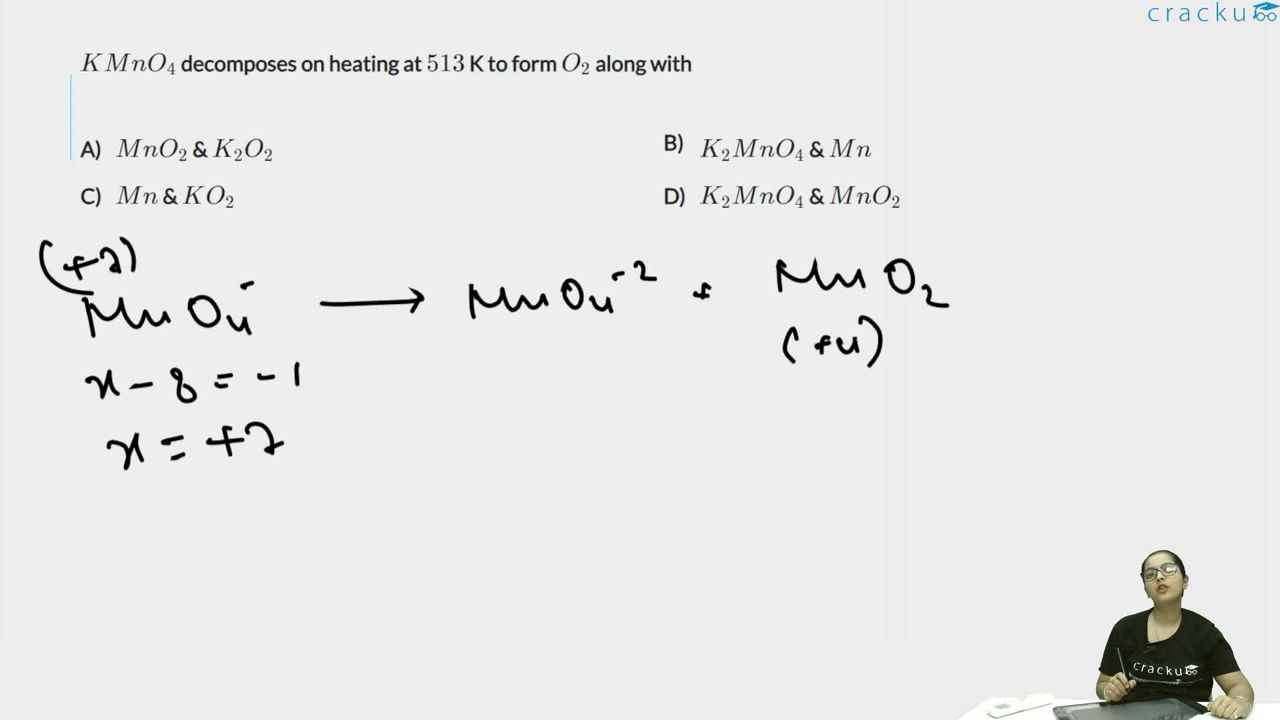
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
