Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
The shortest wavelength of the spectral lines in the Lyman series of hydrogen spectrum is $$915$$ Å. The longest wavelength of spectral lines in the Balmer series will be ______ Å.
Correct Answer: 6588
We are given that the shortest wavelength of the Lyman series is 915 Angstrom, and we need to find the longest wavelength of the Balmer series.
Recall the Rydberg formula for hydrogen spectral lines
The wavelength of spectral lines in hydrogen is given by:
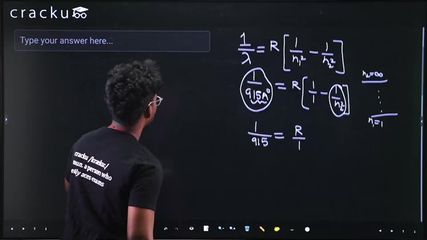
$$\frac{1}{\lambda} = R\left(\frac{1}{n_1^2} - \frac{1}{n_2^2}\right)$$
where $$R$$ is the Rydberg constant, $$n_1$$ is the lower energy level, and $$n_2$$ is the upper energy level.
Find the Rydberg constant from the Lyman series data
The Lyman series corresponds to transitions where $$n_1 = 1$$. The shortest wavelength (series limit) occurs when $$n_2 \to \infty$$:
$$\frac{1}{\lambda_{\infty}} = R\left(\frac{1}{1^2} - \frac{1}{\infty}\right) = R$$
Given $$\lambda_{\infty} = 915$$ Angstrom:
$$R = \frac{1}{915} \text{ per Angstrom}$$
Calculate the longest wavelength of the Balmer series
The Balmer series corresponds to transitions where $$n_1 = 2$$. The longest wavelength (smallest energy transition) occurs when $$n_2 = 3$$ (the closest upper level):
$$\frac{1}{\lambda} = R\left(\frac{1}{2^2} - \frac{1}{3^2}\right) = R\left(\frac{1}{4} - \frac{1}{9}\right)$$
Finding the common denominator:
$$\frac{1}{4} - \frac{1}{9} = \frac{9 - 4}{36} = \frac{5}{36}$$
Therefore:
$$\frac{1}{\lambda} = R \times \frac{5}{36} = \frac{1}{915} \times \frac{5}{36}$$
$$\lambda = \frac{36}{5R} = \frac{36 \times 915}{5} = \frac{32940}{5} = 6588 \text{ Angstrom}$$
The answer is 6588 Angstrom.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
