Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
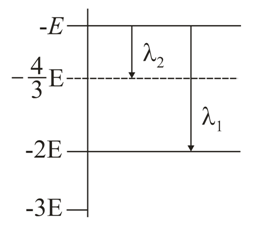
Some energy levels of a molecule are shown in the figure. The ratio of the wavelengths $$r = \frac{\lambda_{1}}{\lambda_{2}}$$, is given by:
$$\Delta E = \frac{hc}{\lambda} \implies \lambda \propto \frac{1}{\Delta E}$$
$$\frac{\lambda_1}{\lambda_2} = \frac{\Delta E_2}{\Delta E_1}$$
For transition $$\lambda_2$$: The transition occurs from level $$-E$$ to level $$-\frac{4}{3}E$$.
$$\Delta E_2 = | -E - (-\frac{4}{3}E) | = | -E + \frac{4}{3}E | = \frac{1}{3}E$$
For transition $$\lambda_1$$: The transition occurs from level $$-E$$ to level $$-2E$$.$$\Delta E_1 = | -E - (-2E) | = | -E + 2E | = E$$
$$r = \frac{\lambda_1}{\lambda_2} = \frac{\Delta E_2}{\Delta E_1}$$
$$r = \frac{\frac{1}{3}E}{E}$$
$$r = \frac{1}{3}$$
Create a FREE account and get:
Predict your JEE Main percentile, rank & performance in seconds

Educational materials for JEE preparation


Ask our AI anything
AI can make mistakes. Please verify important information.
AI can make mistakes. Please verify important information.