Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
A sample of an ideal gas is taken through the cyclic process abca as shown in the figure. The change in the internal energy of the gas along the path ca is -180 J. The gas absorbs 250 J of heat along the path ab and 60 J along the path bc. The work done by the gas along the path abc is:
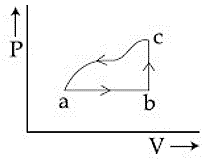
Given:
$$ΔU_{ca}=−180J,\ Q_{ab}=250J,\ Q_{bc}=60J$$
Step 1: Internal energy change along ac
$$ΔU_{ac}=−ΔU_{ca}=−(−180)=180J$$
Step 2: Total heat absorbed along abc
$$Q=Q_{ab}+Q_{bc}=250+60=310J$$
Step 3: Apply First Law of Thermodynamics
Q=ΔU+W
310=180+W
⇒W=130 J
Create a FREE account and get:
Educational materials for JEE preparation
