NTA JEE Mains 24th Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 61
A solution is prepared by dissolving 0.3 g of a non-volatile non-electrolyte solute 'A' of molar mass 60 g $$mol^{-1}$$ and 0.9 g of a non-volatile non-electrolyte solute 'B' of molar mass 180 g $$mol^{-1}$$ in 100 mL $$H_{2}O$$ at 27°C. Osmotic pressure of the solution will be
[Given: R = 0.082 L atm $$K^{-1} mol^{-1}$$]
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 62
Given below are two statements:
Statement I: The number of paramagnetic species among $$[CoF_{6}]^{3-},[TiF_{6}]^{3-},V_{2}O_{5}\text{ and }[Fe(CN)_{6}]^{3-}$$ is 3.
Statement II: $$K_{4}[Fe(CN)_{6}] < K_{3}[Fe(CN)_{6}] < [Fe(H_{2}O)_{6}]SO_{4}.H_{2}O < [Fe(H_{2}O)_{6}]Cl_{3}$$ is the correct order in terms of number of unpaired electron(s) present in the complexes.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 63
Among the following, the CORRECT combinations are
$$\text{A. IF}_{3} \rightarrow \text{T-shaped}(\text{sp}^{3}\text{d})$$
$$\text{B. IF}_{5} \rightarrow \text{Square pyramidal(sp}^{3}\text{d}^{2})$$
$$\text{C. IF}_{7} \rightarrow \text{Pentagonal bipyrnmidal(sp}_{3}\text{d}^{3})$$
$$\text{D. ClO}_{4}^{-} \rightarrow \text{Square planar(sp}^{3}\text{d})$$
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 64
'W' g of a non-volatile electrolyte solid solute of molar mass 'M' g $$mol^{-1}$$ when dissolved in 100 mL water, decreases vapour pressure of water from 640 mm Hg to 600 mm Hg. If aqueous solution of the electrolyte boils at 375 K and $$K_{b}$$ for water is 0.52 K kg $$mol^{-1}$$, then the mole fraction of the electrolyte solute ($$x_{2}$$) in the solution can be expressed as
(Given : density of water= 1 g/mL and boiling point of water= 3 73 K)
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 65
Consider the following two reactions A and B.
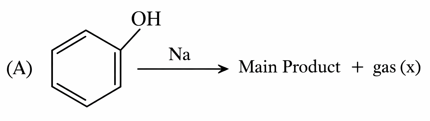
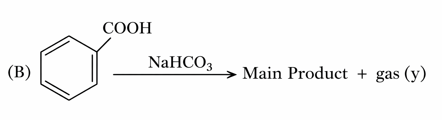
Numerical value of [ molar mass of x + molar mass of y] is __
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 66
Match the LIST-I with LIST-II
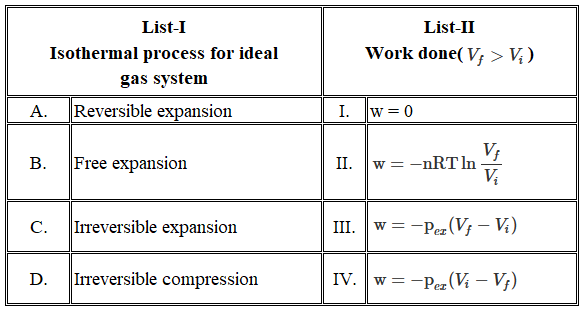
Choose the correct answer from the options given befow:
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 67
A student is given one compound among the following compounds that gives positive test with Tollen's reagent
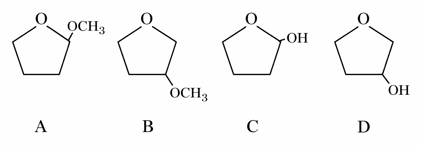
The compound is :
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 68
The correct stability order of the following diazonium salts is
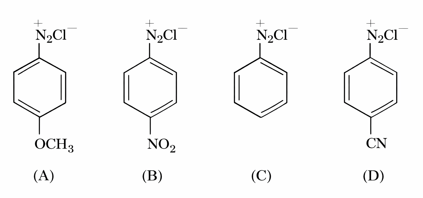
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 69
$$A\rightarrow D$$ is an endothermic reaction occurring in three steps ( elementary).
(i) $$A\rightarrow B \triangle H_{i}=+ve$$
(ii) $$B\rightarrow C \triangle H_{ii}=-ve$$
(iii) $$C\rightarrow D \triangle H_{iii}=-ve$$
Which of the following graphs between potential energy (y-axis) vs reaction coordinate (x-axis) correctly represents the reaction profile of A-> D?
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 70
Match the LIST-I with LIST-II
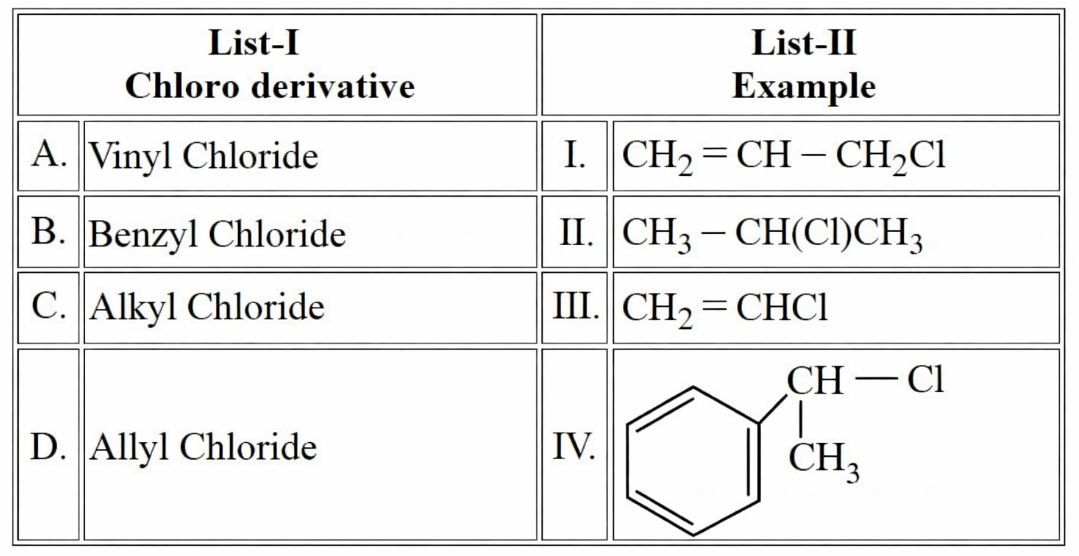
Choose the correct answer from the options given below:


