NTA JEE Mains 24th Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 51
Given below are h¥o statements:
Statement I: Hybridisation, shape and spin only magnetic moment of $$K_{3}[Co(CO_{3})_{3}]$$ is $$sp^{3}d^{2}$$, octahedral and 4.9 BM respectively.
Statement II: Geometty, hybridisation and spin only magnetic moment values (BM) of the ions $$[Ni(CN)_{4}]^{2-},[MnBr_{4}]^{2-}\text{ and }[CoF_{6}]^{3-}$$ respectively are square planar, tetrahedral, octahedral;$$dsp^{2},sp^{3},sp^{3}d^{2}$$ and 0, 5.9, 4.9.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 52
Arrange the following carbanions in the decreasing order of stability.
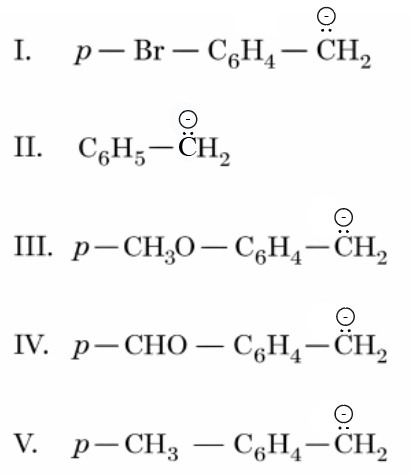
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 53
Consider three metal chlorides x, y and z, where x is water soluble at room temperature, y is sparingly soluble in water at room temperature and z is soluble in hot water. x, y and z are respectively
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 54
At 27 °C in presence of a catalyst, activation energy of a reaction is lowered by $$10 \text{KJ mol}^{-1}$$. The logarithm of ratio of $$\frac{k(\text{catalysed})}{k(\text{uncatalysed})}$$ is......
(Consider that the frequency factor for both the reactions is same)
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 55
A hydroxy compound (X) with molar mass 122 g $$mol^{-1}$$ is acetylated with acetic anhydride, using a large excess of the reagent ensuring complete acetylation of all hydroxyl groups. The product obtained has a molar mass of 290 g $$mol^{-1}$$. The number of hydroxyl groups present in compound (X) is:
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 56
Anange the following alkenes in decreasing order of stability.
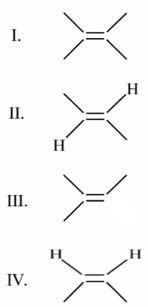
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 57
Given below are two statements:
Statement I: 'C - Cl' bond is stronger in $$CH_{2}=CH-Cl\text{ and }CH_{3}-CH_{2}-Cl$$
Statement II: The given optically active molecule,
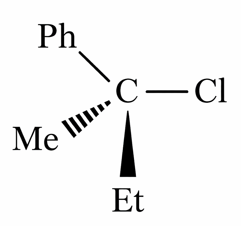
on hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 58
Consider a mixtm-e 'X' which is made by dissolving 0.4 mol of $$[Co(NH_{3})_{5}SO_{4}]Br$$ and 0.4 mol of $$[Co(NH_{3})_{5}Br]SO_{4}$$ in water to make 4 L of solution. When 2 L of mixture 'X' is allowed to react with excess of $$AgNO_{3}$$,it fonns precipitate 'Y'. The rest 2 L of mixture 'X' reacts with excess $$BaCl_{2}$$ to fonn precipitate 'Z'. Which of the following statements is CORRECT?
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 59
Given below are two statements:
Statement I: K > Mg > Al > B is the correct order in terms of metallic character.
Statement II: Atomic radius is always greater than the ionic radius for any element.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Question 60
Given below are statements about some molecules/ions.
Identify the CORRECT statements.
A The dipole moment value of $$NF_{3}$$ is higher than that of $$NH_{3}$$.
B. The dipole moment value of $$BeH_{2}$$ is zero.
C. The bond order of $$O_{2}^{2-}$$ and $$F_{2}$$ is same.
D. The formal charge on the central oxygen atom of ozone is -1 .
E. In $$NO_{2}$$, all the three atoms satisfy the octet rule, hence it is very stable.
Choose the correct answer from the options given below:


