NTA JEE Mains 23rd Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 61
Identify the INCORRECT statements from the following:
A. Notation $$_{12}^{24}Mg$$ represents 24 protons and 12 neutrons.
B. Wavelength of a radiation of frequency $$ 4.5\times10^{15}s^{-1} $$ is $$ 6.7\times10^{-8} $$m.
C. One radiation has wavelength =$$\lambda_1(900nm) $$ and energy= $$E_{1}$$. Other radiation has wavelength = $$\lambda_2(300nm) $$ and energy= $$E2 \cdot E1 : E_2= 3 : 1$$.
D. Number of photons of light of wavelength 2000 pm that provides 1 J of
energy is $$1.006 x 10^{16}$$.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 62
Given below are two statements:
Statement I: $$(CH_3)_3C^+$$ is more stable than $$CH_3^+$$ as nine hyperconjugation interactions are possible in $$(CH_3)_3C^+$$.
Statement II: $$CH_3^+$$ is less stable than $$(CH_3)_3C^+$$ as only three hyperconjugation interactions are possible in $$CH_3^+$$.
In the light of the above statements, choose the correct answer from the options given below
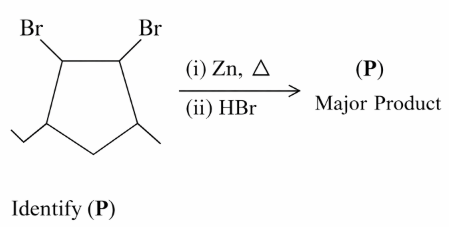
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 64
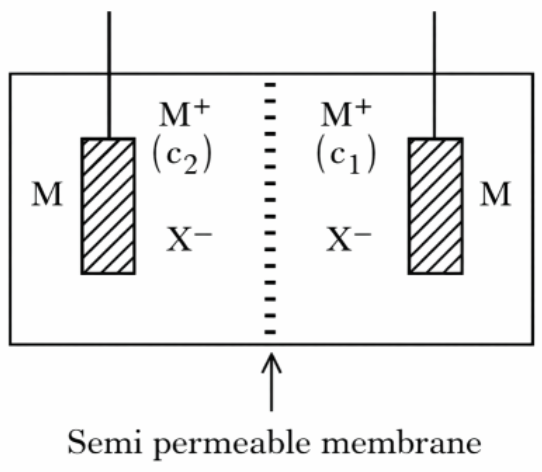
Consider the above electrochemical cell where a metal electrode (M) is undergoing redox reaction by forming $$M^{+}(M\rightarrow M^{+}+e^{-})$$. The cation $$M^{+}$$ is present in two different concentrations $$c_{1}$$ and $$c_{2}$$ as shown above. which of the following statement is correct for generating a positive cell potential?
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 65
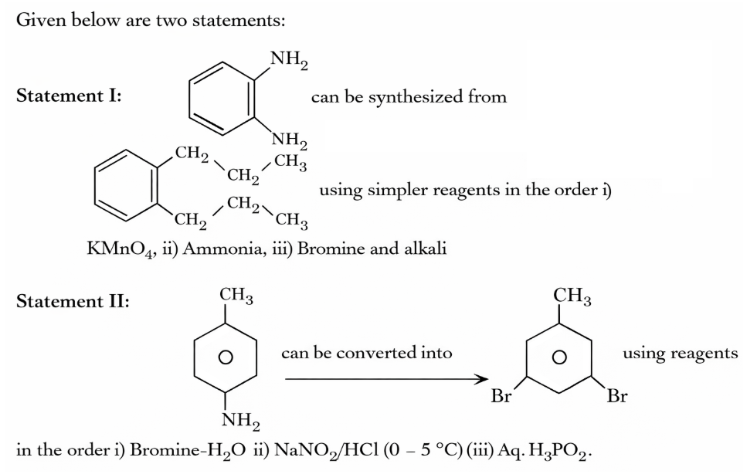
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 66
In Carius method 0.2425 g of an organic compound gave 0.5253 g silver chloride.
The percentage of chlorine in the organic compound is
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 67
Which statements are NOT TRUE about $$XeO_2 F_2$$?
A. It has a see-saw shape.
B. Xe has 5 electron pairs in its valence sheU in XeO 2 F 2.
C. The $$O - Xe- O$$ bond angle is close to $$180^{o}$$.
D. The $$F- Xe -F$$ bond angle is close to $$180^{o}$$.
E. $$Xe$$ has 16 valence electrons in $$XeO_2 F_2$$.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 68
Observe the following reactions at T(K).
I. $$A\rightarrow$$products
II. $$5Br^{-}(aq)+BrO_{3}\text{ } ^{-}(aq)\rightarrow 3Br_2(aq)+3H_2O(l)$$
Both the reactions are started at 10.00 am. The rates of these reactions at 10.10 am are same. The value of $$-\frac{\triangle[Br^{-}]}{\triangle t}$$ at 10.10am. is $$2\times 10^{-4} mol \text{ }L^{-1}min^{-1}$$. The concentration of A at 10.10am is $$10^{-1}mol \text{ } L^{-1}$$. What is the first order rate constant (in min^{-1}) of reaction I?
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 69
Which of the following statements are TRUE about Haloform reaction?:
A. Sodium hypochlorite reacts with KI to give KOI.
B. KOI is a reducing agent.
C. $$ \alpha,\beta$$-unsaturated methylketone $$\mathrm{CH_3 - CH = CH - C(=O) - CH_3}$$ will give iodoform reaction.
D. Isopropyl alcohol will not give iodoform test.
E. Methanoic acid will give positive iodoform test.
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 70
The oxidation state of chromium in the final product formed in the reaction between $$KI$$ and acidified $$K_2 Cr_2 O_7$$ solution is: