NTA JEE Mains 23rd Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 71
$$X_2(g ) + Y_2(g ) \rightleftharpoons 2Z(g)$$
$$X_2(g )$$ and $$Y_2(g )$$ are added to a 1 L flask and it is found that the system attains the above equilibrium at T(K) with the number of moles of $$X_2(g ),\text{ } Y_2(g )$$ and $$Z(g)$$ being 3, 3 and 9 mol respectively (equilibrium moles). Under this condition of equilibrium, 10 mol of $$Z(g$$) is added to the flask and the temperature is maintained at $$T(K)$$. Then the number of moles of $$Z(g)$$ in the flask when the new equilibrium is established is __ . (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 72
Consider the following reaction of benzene.
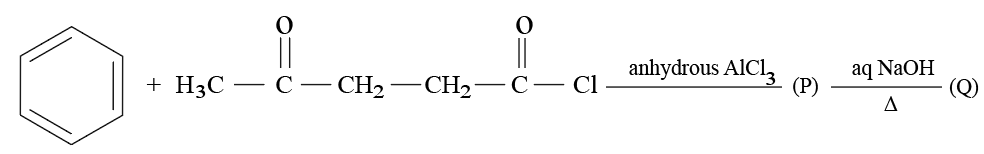
In compound (Q), the percentage of oxygen is ___ %. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 73
200 cc of $$ x\times 10^{-3} M$$ potassium dichromate is required to oxidise 750 cc of 0.6 M Mohr's salt solution in acidic mediun.
Here $$x$$ =___________
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 74
Two liquids A and B form an ideal solution. At 320 K, the vapour pressure of the solution, containing 3 mol of A and 1 mol of B is 500 mm Hg. At the same
temperature, if 1 mol of A is farther added to this solution, vapour pressure of the solution increases by 20 mm Hg. Vapour pressure (in mm Hg) of B in pure state is ____ . (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 75
Total number of unpaired electrons present in the central met al atoms/ions of
$$[Ni(CO)_4],[NiCl_4]^{2-},[PtCl_2(NH_3)_2],[Ni(CN)_4]^{2-}$$ and $$[Pt(CN)_4]^{2-}$$ is_____
789
456
123
0.-
Clear All


