NTA JEE Mains 23rd Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 51
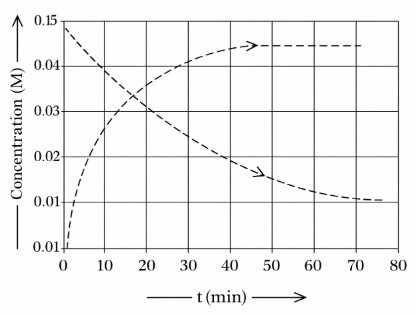
Given above is the concentration vs time plot for a dissociation reaction : $$A \rightarrow nB$$ .
Based on the data of the initial phase of the reaction (initial 10 min), the value of n is________.
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 52
It is noticed that $$Pb^{2+}$$ is more stable than $$Pb^{4+}$$ but $$Sn^{2+}$$ is less stable than $$Sn^{4+}$$
Observe the following reactions.
$$ PbO_2+Pb\rightarrow 2PbO;\triangle_rG^{o}(1) $$
$$ SnO_2+Sn\rightarrow 2SnO;\triangle_rG^{o}(2) $$
Identify the correct set from the following
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 53
Elements X and Y belong to Group 15 . The difference between the electronegativity values of 'X' and phosphorus is higher than that of the difference between phosphorus and 'Y'. 'X' & 'Y' are respectively
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 54
Given below are two statements:
Statement I: The second ionisation enthalpy of Na is larger than the corresponding ionisation enthalpy of Mg.
Statement II: The ionic radius of $$O^{2-}$$ is larger than that of $$F^{-}$$.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 55
Both human DNA and RNA are chiral molecules. The chirality in DNA and RNA arises due to the presence of
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 56
A mixed ether (P), when heated with excess of hot concentrated hydrogen iodide produces two different alkyl iodides which when treated with aq. NaOH give compounds (Q) and (R). Both (Q) and (R) give yellow precipitate with NaOI. Identify the mixed ether (P):
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 57
Identify the CORRECT set of details from the following:
A. $$[Co(NH_3)_6]^{3+}$$ : Inner orbital compex; $$d^{2}sp^{3}$$ hybridized
B. $$[MnCl_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
C. $$[CoF_6]^{3-}$$ : Outer orbital complex; $$d^{2}sp^{3}$$ hybridized
D. $$[FeF_6]^{3-}$$ : Outer orbital complex; $$sp^{3}d^{2}$$ hybridized
E. $$[Ni(CN)_4]^{2-}$$ : Inner orbital complex; $$sp^{3}d^{2}$$ hybridized
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 58
A student has been given a compound "x" of molecular formula- $$C_{6}H_{7}N$$. 'x' is sparingly soluble in water. However, on addition of dilute mineral acid, 'x' becomes soluble in water. 'x' when treated with $$CHCl_{3}$$ and KOH(alc), 'Y' is produced. 'y' has a specific unpleasant smell. On treatment with benzenesulphonyl chloride, 'x' gives a compound 'z' which is soluble in alkali. The number of different "H" atoms present in 'z' is :-
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 59
Iodoform test can differentiate between
A. Methanol and Ethanol
B. $$CH_{3}COOH$$ and $$CH_{3}CH_{2}COOH$$
C. Cyclohexene and cyclohexanone
D. Diethyl ether and Pentan-3-one
E. Anisole and acetone
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 2 - Question 60
The work functions of two metals ($$M_{A}$$ and $$M_{B}$$ ) are in the 1 : 2 ratio. When these metals are exposed to photons of energy 6 eV, the kinetic energy of liberated electrons of $$M_{A}$$ : $$M_{B}$$ is in the ratio of 2.642 : 1. The work functions (in eV) of $$M_{A}$$ and $$M_{A}$$ are respectively.


