NTA JEE Mains 23rd Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 61
Which of the following statements regarding the energy of the stationary state is true in the following one - electron systems?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 62
Consider the general reaction given below at 400 K
$$xA(g)\rightleftharpoons yB(g).
The values of $$K_{p}\text{ and }K_{c}$$ are studied under the same condition of temperature but variation in x and y
(i)$$K_{p}=85.87\text{ and }K_{c}=2.586$$ appropriate units
(ii)$$K_{p}=0.862\text{ and }K_{c}=28.62$$ appropriate units
The values of x and yin (i) and (ii) respectively are:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 63
Compound 'P' undergoes the following sequence of reactions:
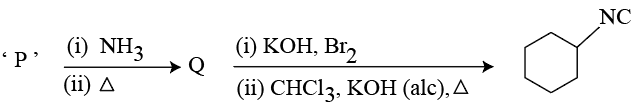
'P' is:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 64
A cup of water at 5°C (system) is placed in a microwave oven and the oven is turned on for one minute during which the water begins to boil. Which of the following option is true ?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 65
ldentify the molecule (X) with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $$HNO_{3},H{2}SO_{4},NF_{3}\text{ and }O_{3}$$. Choose the correct bond angle made by the central atom of the molecule (X).
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 66
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are:
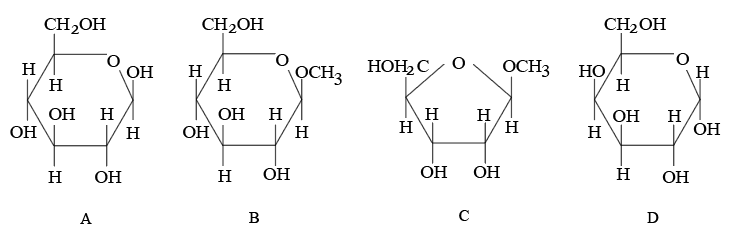
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 67
But-2-yne and hydrogen (one mole each) are separately treated with (i) Pd/C and (ii) Na/ liq. NH3 to give the products X and Y respectively.
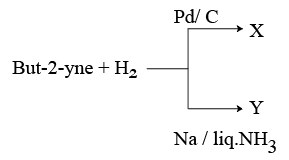
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of X is zero.
C. Boiling point of X is higher than Y.
D. X and Y react with $$O_{3}/Zn+H_{2}O$$ to give different products.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 68
'x' is the product which is obtained &om propanenitrile and stannous chloride in the presence of hydrochloric acid followed by hydrolysis. 'y' is the product which is obtained from the but-2-ene by the ozonolysis followed by hydrolysis. From the followu1g, which product is not obtained when one mole of 'x' and one mole of 'y' react with, each other in the presence of alkali followed by heating?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 69
In the given electrochemical cell, $$Ag(s)|AgCl(s)|FeCl_{2}(aq)$$, $$FeCl_{3}(aq)|Pt(s)$$ at298 K, theceU potential $$E_{cell}$$ will increase when:
A. Concentration of $$Fe^{2+}$$ is increased.
B. Concentration of $$Fe^{3+}$$ is decreased.
C. Concentration of $$Fe^{2+}$$ is decreased.
D. Concentration of $$Fe^{3+}$$ is increased.
E. Concentration of $$Cl^{-}$$ is increased.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 70
Given,
(A) $$n = 5, m_{1} = -1$$
(B) $$n = 3, 1 = 2, m_{1} = -1, m_{2} = +\frac{1}{2}$$
The maximum number of electron(s) in an atom that can have the quantum numbers as given in (A) and (B) respectively are:


