NTA JEE Mains 23rd Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 51
Match List - I with List - II.
.png)
choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 52
The statements that are incorrect about the nickel(II) complex of dimethylglyoxime are:
A. It is red in colour.
B. It has a high solubility in water at pH =9.
C. The Ni ion has two unpaired d-electrons.
D. The N - Ni - N bond angle is almost close to 90°
E. The complex contains four five-membered metallacycles (metal containing rings).
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 53
The correct statements from the following are:
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 54
Consider the following compounds
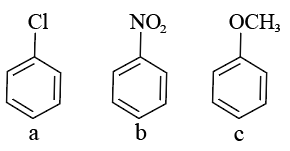
Arrange these compounds in the increasing order of reactivity with nitrating mixture.
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 55
Given below are two statements :
Statement I: $$[CoBr_{4}]^{2-}$$ ion will absorb light of lower energy than $$[CoCl_{4}]^{2-}$$ ion.
Statement II: In $$[Col_{4}]^{2-}$$ ion, the energy separation between the two set of d-orbitals is more than $$[CoCl_{4}]^{2-}$$ ion.
ln the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 56
Which one of the following graphs accurately represents the plot of partial pressure of $$CS_{2}$$ vs its mole fraction in a mixture of acetone and $$CS_{2}$$ at constant temperature?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 57
Given below are two statements :
Statement I: Sublimation is used for the separation and purification of compounds with low melting Point.
Statement II: The boiling point of a liquid increases as the external pressure is reduced.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 58
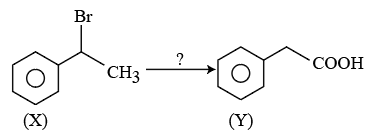
The correct sequence of reagents for the above conversion of X to Y is:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 59
Consider the following sequence of reactions.
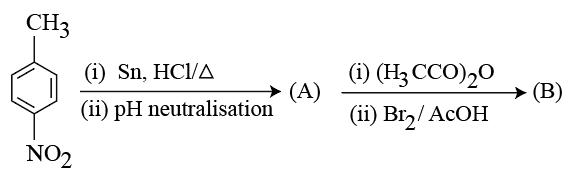
4-Nitrotoluene
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce ____ mg of B.
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N: 14, 0: 16, Br : 80)
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 60
The correct trend in the first ionization enthalpies of the elements in the $$3^{rd}$$ period of periodic table is:


