Sign in
Please select an account to continue using cracku.in
↓ →

Join Our JEE Preparation Group
Prep with like-minded aspirants; Get access to free daily tests and study material.
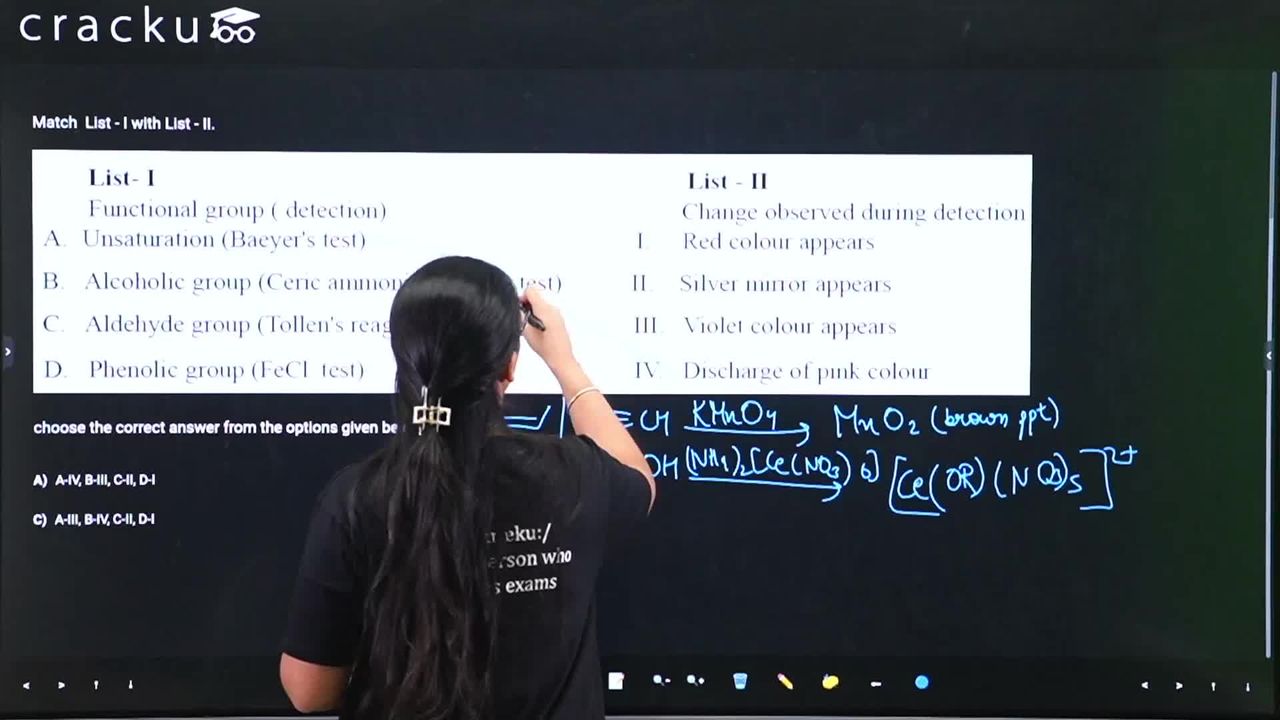
Match List - I with List - II.
.png)
choose the correct answer from the options given below :
Baeyer's reagent is a cold, alkaline solution of potassium permanganate ($$\text{KMnO}_4$$), which is deep pink/purple. When it reacts with an alkene or alkyne, the $$\text{MnO}_4^-$$ ion is reduced to $$\text{MnO}_2$$ (a brown precipitate), causing the pink colour to disappear (decolourise).
Ceric ammonium nitrate ($$\text{CAN}$$) is an orange-yellow reagent. When added to an alcohol, it forms a alkoxy cerium(IV) complex, which results in a characteristic wine-red colouration.
Tollen's reagent contains the silver-ammonia complex $$[\text{Ag}(\text{NH}_3)_2]^+$$. Aldehydes reduce these silver ions to metallic silver, which deposits on the inner walls of the test tube, creating a shining silver mirror.
Phenols react with neutral ferric chloride to form a coordination complex with the iron. For simple phenol, this complex is typically violet or purple, though other phenols can produce green or blue colours.
Click on the Email ☝️ to Watch the Video Solution
Create a FREE account and get:
Educational materials for JEE preparation
