NTA JEE Mains 23rd Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 71
The crystal field splitting energy of $$[Co(oxalate)_{3}]^{3-}$$ complex is 'n' times that of the $$[Cr(oxalate)_{3}]^{3-}$$ complex. Here 'n' is_______. (Assume d $$\triangle_{\circ} > > P$$)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 72
For the following gas phase equilibrium reaction at constant temperature,
$$NH_{3}(g)\rightleftharpoons 1/2N_{2}(g)+3/2H_{2}(g)$$
if the to tal pressure is $$\sqrt{3}$$ atm and the pressure equilibrium constant ($$K_{p}$$) is 9 atm, then the degree of dissociation is given as $$(x\times 10^{-2})^{-1/2}$$.The value of x is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 73
x mg of pure HCl was used to make an aqueous solution. 25.0 mL of 0.1 M $$Ba(OH)_{2}$$ solution is used when the HCl solution was titrated against it. The numerical value of x is ______$$\times 10^{-1}$$. (nearest integer)
Given : Molar mass of HCl and $$Ba(OH)_{2}$$ are 36.5 and 171.0 g $$mol^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 74
Consider all the structural isomers with molecular formula $$C_{5}H_{11}Br$$ are separately treated with KOH(aq) to give respective substitution products, without any rearrangement. The number of products which can exhibit optical isomerism from these is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Question 75
For the thermal decomposition of reactant AB(g), the following plot is constructed.
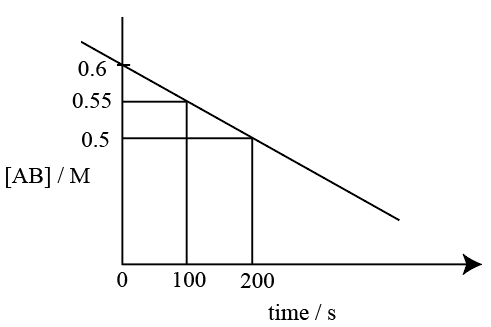
The half life of the reaction is 'x' min.
x= ____ min. (Nearest integer)
789
456
123
0.-
Clear All


