NTA JEE Mains 22nd Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 61
Identify the number of structure/s from the following which can be correlated to D-glyceraldehyde.
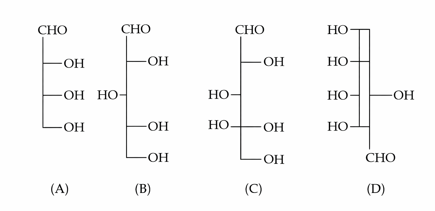
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 62
Given below are two statements : Statement (I) : A spectral line will be observed for a $$2p_{x}\rightarrow 2p_{y}$$ transition.Statement (II) :$$2p_{x}$$ and $$2p_{y}$$ are degenerate orbitals. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 63
Given below are two statements : Statement (I) : An element in the extreme left of the periodic table forms acidic oxides. Statement (II) : Acid is formed during the reaction between water and oxide of a reactive element present in the extreme right of the periodic table. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 64
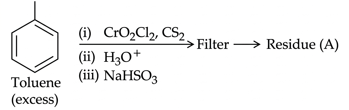
Residue (A) + HCl (dil)$$\rightarrow $$ Compound (B) Structure of residue (A) and Compound (B) formed respectively is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 65
The alkane from below having two secondary hydrogens is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 66
When sec-butylcyclohexane reacts with bromine in the presence of sunlight, the major product is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 67
The most stable carbocation from the following is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 68
Match the Compounds (List - I) with the appropriate Catalyst/Reagents (List - II) for their reduction into corresponding amines.
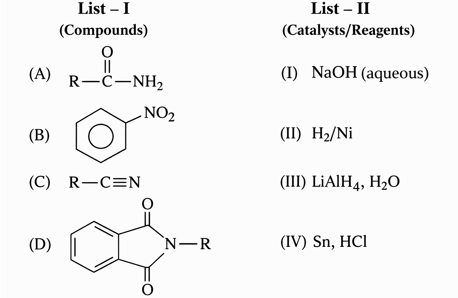
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 69
Arrange the following compounds in increasing order of their dipole moment : $$HBr,H_{2}S,NF_{3}\text{ and }CHCl_{3}$$
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 70
The maximum covalency of a non-metallic group 15 element ' E ' with weakest E-E bond is:


