NTA JEE Mains 22nd Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 51
Given below are two statements : Statement (I) : Nitrogen, sulphur, halogen and phosphorus present in an organic compound are detected by Lassaigne's Test. Statement (II) : The elements present in the compound are converted from covalent form into ionic form by fusing the compound with Magnesium in Lassaigne's test. In the light of the above statements, choose the correct anower from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 52
Density of 3 M NaCl solution is $$1.25 g/mL$$. The molality of the solution is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 53
The correct order of the following complexes in terms of their crystal field stabilization energies is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 54
Given below are two statements : Statement (I) : Corrosion is an electrochemical phenomenon in which pure metal acts as an anode and impure metal as a cathode. Statement (II) : The rate of corrosion is more in alkaline medium than in acidic medium. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 55
Consider the given figure and choose the correct option :
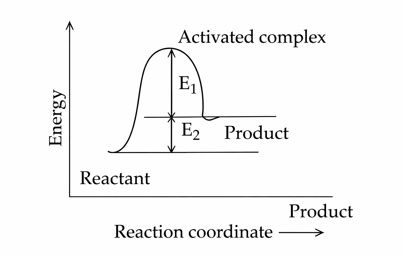
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 56
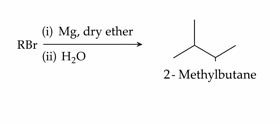
The maximum number of RBr producing 2-methylbutane by above sequence of reactions is ________ -
(Consider the structural isomers only)
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 57
The species which does not undergo disproportionation reaction is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 58
The molar solubility(s) of zirconium phosphate with molecular formula $$(Zr^{4+})_{3}(PO_{4}^{3-})_{4}$$ is given by relation :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 59
Identify the homoleptic complex(es) that is/are low spin. $$(A)[Fe(CN)_{5}NO]^{2-}(B)[CoF_{6}]^{3-}(C)[Fe(CN)_{6}]^{4-}(D)[Co(NH_{3})_{6}]^{3+}(E)[Cr(H_{2}O)_{6}]^{2+}$$ Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Question 60

Match List - I with List - II.
Choose the correct answer from the options given below :


