NTA JEE Mains 22nd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 51
Radius of the first excited state of Helium ion is given as : $$a_0\rightarrow$$ radius of first stationary state of hydrogen atom.
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 52
The incorrect statements regarding geometrical isomerism are : (A) Propene shows geometrical isomerism. (B) Trans isomer has identical atoms/groups on the opposite sides of the double bond. (C) Cis-but-2-ene has higher dipole moment than trans-but-2-ene. (D) 2-methylbut-2-ene shows two geometrical isomers. (E) Trans-isomer has lower melting point than cis isomer. Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 53
A liquid when kept inside a thermally insulated closed vessel $$25^{o}C$$ at was mechanically stirred from outside. What will be the correct option for the following thermodynamic parameters ?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 54
Which of the following electronegativity order is incorrect?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 55
Lanthanoid ions with $$4f^{7} configuration are : $$(A)Eu^{2+}$$\text{ }$$ (B)Gd^{3+}$$\text{ }$$ (C)Eu^{3+}$$\text{ }$$ (D)Tb^{3+}$$\text{ }$$ (E)Sm^{3+}$$ Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 56
Given below are two statements : Statement I : One mole of propyne reacts with excess of sodium to liberate half a mole of $$H_2 $$ gas. Statement II : Four g of propyne reacts with $$ NaNH_2 \text{ to liberate } NH_3$$ gas which occupies 224 mL at STP. In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 57
The compounds which give positive Fehling's test are :
(A).
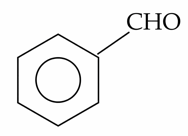
(B).
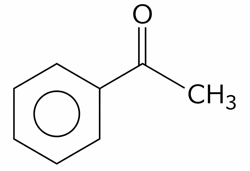
(C). $$ HOCH_{2} - CO - (CHOH)_{3}-CH_{2}-OH $$
(D).
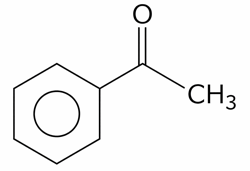
(E).
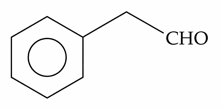
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 58
Which of the following electrolyte can be used to obtain $$H_2 S_2 O_8 $$ by the process of electrolysis?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 59
Given below are two statements : Statement I : $$ CH_3 - O - CH_2 - Cl$$ will undergo $$S_N 1$$ reaction though it is a
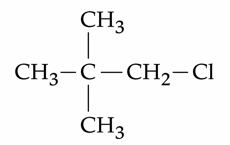
primary halide. Statement II :
will not undergo $$S_N 2$$ reaction very easily though it is a primary halide. In the light of the above statements,
choose the most appropriate answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 60
Which of the following acids is a vitamin?


