NTA JEE Mains 22nd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 71
The number of molecules/ions that show linear geometry among the following is ______
$$\mathrm{SO_2}$$, $$\ \mathrm{BeCl_2}$$, $$\ \mathrm{CO_2}$$, $$\ \mathrm{N_3^-}$$, $$\ \mathrm{NO_2}$$, $$\ \mathrm{F_2O}$$, $$\ \mathrm{XeF_2}$$, $$\ \mathrm{NO_2^+}$$, $$\ \mathrm{I_3^-}$$, $$\ \mathrm{O_3}$$
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 72
$$ A \rightarrow B $$ The molecule A changes into its isomeric form B by following a first order kinetics at a temperature
of 1000 K . If the energy barrier with respect to reactant energy for such isomeric transformation is $$191.48kJ$$ $$Mol^{-1}$$ and the frequency factor is $$10^{20},$$ the time required for 50% molecules of A to become B is
_________ picoseconds (nearest integer). $$[R=8.314 JK^{-1} mol^{-1}]$$
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 73
Consider the following sequence of reactions :
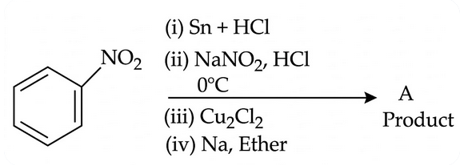
Molar mass of the product formed (A) is ______$$gmol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 74
Some $$CO_2$$ gas was kept in a sealed container at a pressure of 1 atm and at 273 K . This entire amount of $$CO_2$$ gas was later passed through an aqueous solution of $$Ca(OH)_2$$. The excess unreacted $$Ca(OH)_2$$ was later neutralized with 0.1 M of 40 mL HCl . If the volume of the sealed container of $$CO_2$$ was $$x$$, then $$x$$ is ________$$cm^{3}$$ (nearest integer). [Given : The entire amount of $$CO_2 (g)$$reacted with exactly half the initial amount of $$Ca(OH)_2$$ present in the aqueous solution.]
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 75
In Carius method for estimation of halogens, 180 mg of an organic compound produced 143.5 mg of AgCl . The percentage composition of chlorine in the compound is _______ \%. (Given : molar mass in $$gmol^{-1} \text{ of } Ag: 108, Cl: 35.5$$)
789
456
123
0.-
Clear All


