NTA JEE Mains 22nd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 61
Match List-I with List-II.
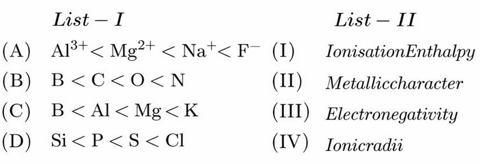
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 62
Which of the following statement is not true for radioactive decay?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 63
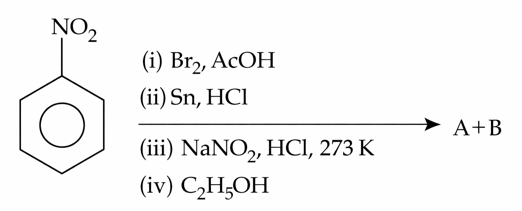
The products formed in the following reaction sequence are :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 64
How many different stereoisomers are possible for the given molecule?
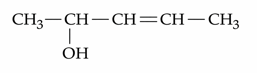
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 65
A vessel at 1000 K contains $$CO_2$$ with a pressure of 0.5 atm . Some of $$CO_2$$ is converted into CO on addition of graphite. If total pressure at equilibrium is 0.8 atm , then Kp is :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 66
A solution of aluminium chloride is electrolysed for 30 minutes using a current of 2 A . The amount of the aluminium deposited at the cathode is [Given : molar mass of aluminium and chlorine are 27g $$mol^{-1} $$ and 35.5 g $$mol^{-1}$$ respectively. Faraday constant $$=96500C mol^{-1}$$ ]
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 67
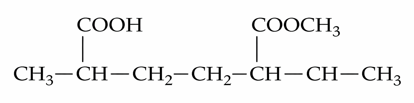
The IUPAC name of the following compound is :
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 68
In which of the following complexes the CFSE, $$\triangle_o$$ will be equal to zero?
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 69
Arrange the following solutions in order of their increasing boiling points. (i) $$10^{-4} M$$ $$NaCl$$ (ii) $$10^{-4}M$$ $$Urea$$ (iii) $$10^{-3} M$$ $$NaCl$$ (iv) $$10^{-2} M$$ $$NaCl$$
NTA JEE Mains 22nd Jan 2025 Shift 1 - Question 70
From the magnetic behaviour of $$[NiCl_4]^{2-} \text{(paramagnetic) and } [Ni(CO)_4] $$ (diamagnetic), choose the correct geometry and oxidation state.


